Why It Matters That Seed Oils Are Being Recognized as Ultra-Processed The US Dietary Guidelines…
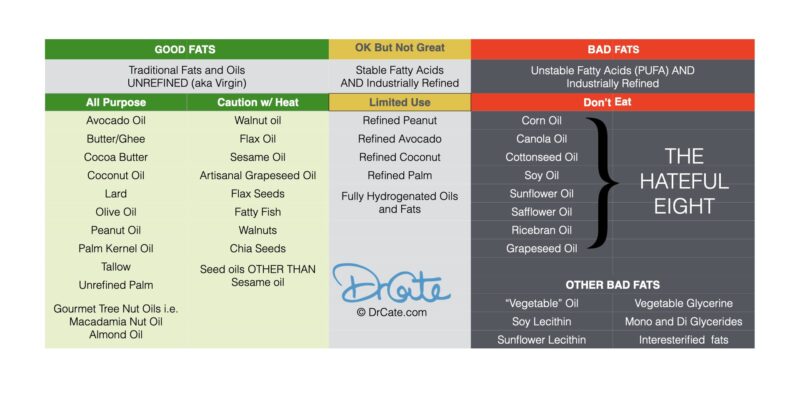
Good Fats vs Bad Fats: The Truth About Seed Oils
Table of Contents
- Bad Fats: NEVER eat these!
- THE HATEFUL EIGHT REFINED PUFA-Rich seed OILS a.k.a. “Vegetable oils”
- Other Franken Fats
- Good Fats: Eat THESE instead!
- Refined Oils: Limited Use
- WHY REFINED VEGETABLE OILS (aka The Hateful Eight Seed Oils) ARE TOXIC BUT THE SEEDS THEY CAME FROM ARE NOT TOXIC
- PUFA Fatty Acids Are Also Nutrients
- LOOKING FOR REFERENCES LINKING SEED OILS TO HEALTH HARMS? Click HERE
- Read this before taking any oil supplement, including fish oil:
- How much vegetable oil is too much?
- THE MYTH OF EXPELLER PRESSED
- COOKING TIPS FOR HIGH HEAT:
- SMOKE POINT
- HOW TO SUBSTITUTE VEGETABLE OIL FOR HEALTHY FATS
- HOW SEED OILS ARE MADE: Refining steps that damage PUFAs
- WHY GOOD FATS GO BAD: THE TWO CAUSES OF RANCID TASTE
- Is eating rancid food unhealthy?
- BEWARE OF “NEUTRAL” FLAVOR
- Harvard Gets It Wrong
- Common scientific terms related to fats and seed oils:
- I read seed oils were bad because of the omega-6, why didn’t you mention that?
- What is artisanal oil and why is it the best?
- Can I eat seeds, nuts or fish that are high in PUFA?
- I heard peanut oil was high in PUFA, why is it listed as good?
- Sesame oil is also high in PUFA, why do you list it as good?
- Flax oil is high in PUFA, why is that good?
- Pumpkin oil, good or bad?
- What about high-oleic sunflower oil, is it better?
- What about soy or sunflower lecithin?
- What are your thoughts on Thrive Algae Oil?
- What about skin products made with seed oils?
- Palm oil is showing up in more foods. How can I tell if it’s refined?
- Is MCT oil acceptable? It is derived from coconut oil, but distilled with heat and pressure. Is it toxic?
- How long does it take to get the excessive PUFA out of my body?
- Sources:
This page is YOUR resource list of all things pertaining to edible fats and oils, please bookmark if you are interested in this topic.
Please read the FREQUENTLY ASKED QUESTIONS section for oils not addressed in the above graphic, and if not there either, then please visit this page where I’ve answered dozens more questions.
To discover how seed oils and other nutritional issues may have affected your health and how to recover the fastest, please schedule a consultation.
This article is continued below...(scroll down)
Bad Fats: NEVER eat these!
THE HATEFUL EIGHT REFINED PUFA-Rich seed OILS a.k.a. “Vegetable oils”
These are the worst of the worst:
Corn, Canola (also called “Rapeseed”), Cottonseed, Soy, Sunflower, Safflower, Grapeseed, Rice bran.
Vegetable oil is an industry term that sometimes includes olive oil, peanut, and coconut. However, when you see the word vegetable oil on the ingredients, it’s not going to be first press, unrefined olive, peanut, or coconut. It will be one of the cheap, refined PUFA-rich, seed oils listed above.
Other Franken Fats
Interesterified fat, hydrogenated fat, trans fat (these are mostly gone from the food supply). These are generally made from the hateful eight.
Good Fats: Eat THESE instead!
Animal Fats & Virgin Oils
Avocado oil, Butter, Coconut Oil, Duck Fat, Ghee, Lard, Olive oil, Peanut oil, Tallow, Sesame oil, Flax oil, Walnut oil, Almond oil, Macadamia nut oil
Anything that says cold-pressed and unrefined. It must say unrefined! If it says cold-pressed but is refined, it’s not good.
USE THESE FOR COOKING:
Almond oil, Avocado oil, Butter, Coconut, Duck Fat, Ghee, Lard, Macadamia nut oil, Peanut oil, Tallow, Sesame/Toasted Sesame oil (Sesame is better if combined with peanut or other stabler fat, details below)
DON’T COOK WITH THESE
Flax oil, Walnut oil, Fish oil
Refined Oils: Limited Use
Not all refined oils are toxic. Refined olive, avocado, peanut, coconut, and palm are in my “ok but not great” category. They are not toxic because they can handle the refining process without oxidizing.
However, they are not going to nourish us. Refining strips away most of the nutrients, like phospholipids, phytosterolsls, antioxidants, vitamins, and minerals.
You can think of these “ok but not great” refined oils as the empty calories of the fat world. If you can’t afford the Good Fats, these are still far better than the Hateful Eight. If you do consume these empty-calorie refined oils, it’s especially vital to ensure that the rest of your calories are nutrient-rich.
WHY REFINED VEGETABLE OILS (aka The Hateful Eight Seed Oils) ARE TOXIC BUT THE SEEDS THEY CAME FROM ARE NOT TOXIC
(The short version)
The seeds used to make the Hateful Eight oils contain a high percentage of polyunsaturated fatty acids (PUFAs). PUFAs are unstable and break down into toxic molecules (like aldehydes) when exposed to oxygen. Because of this, bottles of seed oils contain small amounts of toxins when they leave the factory. But that’s just the beginning.
PUFAs also tend to polymerize. That means a small amount of toxic aldehyde can trigger chain reactions that lead to much larger amounts of toxic aldehyde developing. This happens most rapidly during the cooking process. And it can also happen in our bodies, starting with where they enter our bodies–our gut.
Would You Prefer Tallow Fries or Varnish Fries?
Carpenters use varnish to finish wood. Varnish is often made from vegetable oils, including soy and linseed (which is rich in omega-3, like canola), because these oils are chiefly composed of PUFAs. PUFAs react with oxygen in the air to help polymerize the varnish into a nice, hard coating that helps preserve the wood.
Varnish is good for your floors, but not so good for your gut. The Hateful Eight oils can cause peroxidation in the gut, and that triggers inflammation.
The harms of vegetable oils extend well beyond the gut. Everywhere they go, the same chemistry applies, so they end up triggering inflammation in every type of tissue in every organ in our bodies.
All of my books discuss the connections between the Hateful Eight seed oils, oxidation, inflammation, and disease.
PUFA Fatty Acids Are Also Nutrients
A little bit of PUFA is not a problem for us, we actually need some. And when we get PUFA from whole foods like sunflower, chia or flax seeds, it’s well protected by antioxidants nature builds into the seed. These protectants get stripped away during the industrial-scale refining of sunflower and the other vegetable oils, and that’s just the beginning of the problems with vegetable oils.
By turning soy and sunflower into refined oil, most of the good nutrients the farmer worked hard to grow are removed. But the story gets worse.
The refining process not only strips away antioxidants, but it also oxidizes some of the PUFA. The two main types of PUFA in our oilseeds are omega-6 and omega-3. The refining chemically transforms these would-be nutrients into a wide variety of potent toxins with long names like 4-hydroxynonanal and 4-hydroxyhexanol (both of those are types of aldehydes). As they leave the factory, bottles of soy, canola, and the other Hateful Eight already contain trace amounts of these harmful toxins. And because the protective antioxidants are gone, there’s nothing to stop more toxins from forming.
More information on the refining under “See How it’s Made,” below.
LOOKING FOR REFERENCES LINKING SEED OILS TO HEALTH HARMS? Click HERE
The above link takes you to a semi-organized list of just a few of the hundreds of references I’ve used in my books. There are hundreds more yet to be compiled into the document. If anyone wants to volunteer to help out with this project, please let me know!
Read this before taking any oil supplement, including fish oil:
Supplement companies love extracting oils from whole foods and encapsulating them, and then promoting them as somehow superior to the whole food. Common examples of supplement oils include:
- Fish oil
- Cod liver oil
- Oregano
- Cumin
- Hemp seed
- Flax
- Udo’s oil
- Borage oil
Should you even take fish oil supplements? https://drcate.com/should-i-take-fish-oil-supplements-benefits-and-harms/
Omega-3 supplements are generally not needed on a whole foods diet. Whole foods that are naturally high in omega-3 include raw or sprouted nuts and seeds, pasture-fed dairy and meats, liver (yum) and oily fish. If you are a vegan who cannot eat raw or sprouted nuts and seeds, then a flax oil supplement is a good way to go.
How much vegetable oil is too much?
That’s a little like asking how many cigarettes should a 4 year old smoke. More than none is too much. However because these oils are now added to spice mixes that are added to many otherwise healthy foods, it has become almost impossible to avoid entirely. Other than spice mixes, products with vegetable oil are best avoided whenever possible.
FATS v OIL
Fats are solid at room temperature and oils are liquid. Saturated fats are stiff, so highly saturated fat. (Figure below)
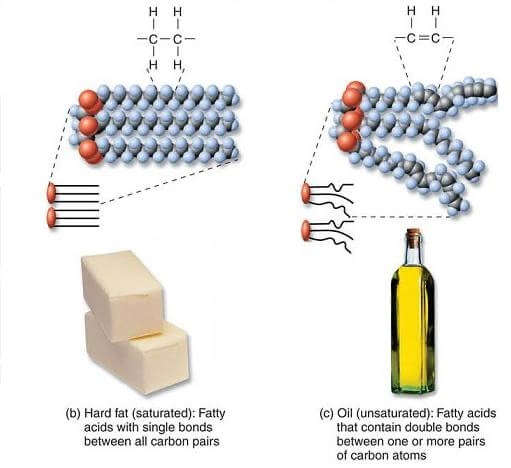
FATTY ACIDS v FATS
Fatty acid refers to a molecule composed of a several carbons linked together, generally anywhere from 4 to 26 carbon atoms, with a special group at the end called a carboxyl. The chain of carbon atoms may be linked together with single bonds, and be saturated, or contain one double bond, and be mono unsaturated, or contain two or more double bonds, and be polyunsaturated. Our bodies cell membranes are composed of all three types of fatty acids. We cannot make certain fatty acids, those have to come from food, these we call essential fatty acids and they come in two types: omega-3 and omega-6.
Fats Most fats and oils we eat are composed of three fatty acids bound to glycerol to form a structure called a triglyceride. Triglycerides are very large molecules and the general idea is a little like three keys dangling off a keychain. When we eat any fat or oil, our digestive system breaks down the triglyceride into free fatty acids and glycerol so that the molecules can get into our intestinal cells, then the intestinal cell reassembles them back into triglyceride and ships them out in the bloodstream as chylomicrons, a kind of lipoprotein. When we eat too much, we store the extra as fat under our skin, all in the form of triglyceride.
THE MYTH OF EXPELLER PRESSED
If my oil says expeller pressed, does that mean its okay?
No. Here’s why:
Bottles of organic oil often state “expeller pressed” as a selling point, to suggest that it has been gently treated, in an extra-virgin sort of way. But that couldn’t be farther from the truth. Firstly, expeller pressed simply means that the first step of the extraction was mechanical. The second step was probably the standard, solvent extraction using hexane.
But once the expeller-pressed oil has been extracted, it’s generally also refined, bleached and deodorized. These three additional treatments guarantee that the polyunsaturated fatty acid molecules will be oxidized in ways that generate toxins like 4-hydroxyhexanal, 4-hydroxynonanol, aldehydes and more. These compounds aren’t just hard to pronounce, they’re hard for our cells to tolerate and lead to mitochondrial uncoupling, DNA damage, free radical cascades and other cell-damaging events that accelerate the aging process and contribute to disabling disease. Don’t be fooled. (For more information, please read chapters 7 and 8 of the 2017 edition of Deep Nutrition: Why Your Genes Need Traditional Food)
COOKING TIPS FOR HIGH HEAT:
STIR FREQUENTLY:
The higher the heat, the more you need to be stirring unless you’re going for a specific effect, like char flavor or crispy skin.
HEALTHY OIL COMBINATIONS:
BUTTER+OLIVE: Add a pat of butter to olive oil when cooking at high heat, the saturated fat in the butter protects the olive oil and the antioxidants in the olive oil protect the protein in the butter that might otherwise burn.
SESAME+PEANUT: Add sesame to peanut oil for Asian dishes. The ratio should be roughly 4-8:1 Peanut:Sesame. Sesame is high in PUFA, but it has powerful antioxidants that, when added to low PUFA peanut oil, protect all the PUFAs.
SMOKE POINT
Should I make sure to use a high smoke point oil for pan frying, wok cooking or other high-heat applications?
No. Here’s why:
Smoke point is a sciencey sounding selling point that vegetable oil salesmen use to ooze their way into busy restaurants. If you’ve read about smoke points, you’ve probably read something like this “Refined oils have higher smoke points and typically a more neutral flavor than unrefined oils, which makes them better for sautéing, frying or even deep-frying.” I think the concept of smoke point is bunk. First of all, what chef is going to literally wait for food on the stove to start smoking before stirring it? Have you ever seen that on a cooking show? Secondly, and this is the more important point, the molecular degradation that occurs in these high smoke point oils both during their manufacture and then again when they’re exposed to high heat during cooking invisibly degrades the oil, generating molecules that are dangerous to our health.
If the food you order has black char on it, you’ll probably realize someone in the kitchen wasn’t paying attention to your dish, and send it back. The higher smoke point oils enable chefs to stir less often and in so doing to overheat your food without leaving any evidence.
I’m not saying theres no such thing as smoke point. Of course there is. But the myth is that the product is somehow superior because it has a high smoke point. You can increase the smoke point of any fat by removing proteins, antioxidants, and free fatty acids. For example, ghee has a higher smoke point than butter because the clarification process reduces the protein content.
I recommend using high-quality oils and fats like butter, lard or tallow, and yes, even EVOO, for stovetop frying. But be sure to stir! It should go without saying that overcooking your dishes not a healthy practice. Who needs high smoke points? Just eat properly cooked food.
HOW TO SUBSTITUTE VEGETABLE OIL FOR HEALTHY FATS
If a recipe calls for vegetable oil you can just about always swap it out. I can’t actually think of any exception at the moment.
Baking: You can use melted butter, coconut oil, or peanut oil (often changes the flavor in a very pleasant way) or any of the “ok but not great” refined oils becuase they will have a neutral flavor.
Dressings: Pick an oil that matches the flavor profile of the cuisine. For example, olive for Italian, French or anything Mediterranean, peanut or sesame (or both) for anything Asian. Or a neutral flavored, refinned oil from the “Ok but not great list” above.
Stovetop cooking. Same rule as with dressings.
HOW SEED OILS ARE MADE: Refining steps that damage PUFAs
STEP 1) EXTRACT CRUDE OIL FROM THE SEED
Manufactures produce a crude oil by extracting it in one of three ways:
- Mechanical extraction, either cold pressed (always below 120 degrees) or expeller pressed (the pressure is higher, which increases the temperature). This is the best. But it leaves a lot of oil behind in the seed or fruit, roughly half for expeller pressing, or more for cold pressing, so it’s typically done only by small batch, artisanal producers. Extra virgin olive oil is produced by mechanical extraction, as are other high-quality oils. The highest quality oils do not need to be refined and so the bottles may contain some cloudy-appearing material that’s actually very good for you. (The video below of Figone’s Olive Oil Pressing at Factory is a good example of what today’s mechanical, expeller pressing system looks like on a relatively small scale.)
- Double extraction. This is probably how most “expeller pressed” oil is actually produced. The manufacturer will mechanically press it first, obtaining about half of the oil they will ultimately be able to extract from the seed. This produces oil and an oil-rich seed cake. To remove the other half of the oil from the seed cake, they process with hexane, as below, making for a more “cost effective” product. Unfortunately, from a consumer choice standpoint, its not clear that oils produced this way won’t be called cold pressed or expeller pressed, even though that’s only part one of the process. It appears there’s simply no way to distinguish oils that have been only mechanically pressed from these “double extraction” oils, where half has been mechanically extracted and half has been processed by solvent extraction, as below.
- Solvent extraction, using hexane (also in your gasoline tank). This is the worst. They do remove as much of the hexane as they can, using bleaches or distillation, and it’s not quite as damaging to the polyunsaturated oil molecules as the refining process.
What is the best oil? See the heading in the FAQ section below: What is artisanal oil?
STEP 2) DEGUMMING
After the double extraction and solvent extraction, a sticky soapy sludge develops on top of the oil as a result of churning the phosphatides (when you work soap into a lather, your churning the phosphatides with water creating the foam). These phosphatides must be removed before refining, and so manufacturers use a “wash” of sodium hydroxide and water to accomplish this.
Cold pressed oils do not generally require degumming
STEP 3) REFINE the CRUDE OIL
Step one and, if it’s needed, step two produce what’s called a “crude oil.” Other than cold pressed oils, the crude oils all contain numerous contaminants you would not want to eat. So the manufacturer cleans it up in three more very harsh steps that damage the PUFAs: refining, bleaching and deodorizing.
Refining: This is performed to remove the free fatty acids, which would contribute to a rancid taste. This is accomplished with either an acid or bleaching agent. In the latter case, the since bleaching has been performed the process skips ahead to deodorizing.
Bleaching: This is performed to remove chlorophyll, the chemical in plants that imparts the green color. Chlorophyll must be removed from these high PUFA seed oils because it promotes rapid oxidation of the PUFA fatty acids and would lead to a very sludgy, sticky oil that wouldn’t pour out of the bottle very well. While bleaching improves its pour-ability, it also generates partially oxidized PUFAs compounds. These are the highly toxic compounds that promote oxidative stress in our bodies and can damage our DNA.
Deodorizing: This is performed to remove flavor components, which would come from chemicals originally present in the seed (such as antioxidant phytonutrients) as well as byproducts of the above steps. This is performed by heating the oil again to 510 F/ 265 c and forcing steam through it to try to capture the volatile materials.
FURTHER PROCESSING:
The refined oil can be further treated to raise the melting point to create the desired, more solid texture. One method is hydrogenation, which creates a partially hydrogenated, solid fat. Another is inter-esterification, which rearrange the fatty acid locations on glycerol and also solidifies the fat. Both of these lead to generation of different forms of toxins than the above. Hydrogenation leads to the generation of fatty acids with a single trans bond, which block our body’s enzymes. Interesterification leads to the formation of triglycerides with unusual configurations and has been found to lead to elevated blood glucose levels.
WHY GOOD FATS GO BAD: THE TWO CAUSES OF RANCID TASTE
Nature does not make bad fats, factories do. By mass producing oils, we can damage them in two different ways that can lead to rancid taste.
- ENZYMES. When oils and fats are too old, they can break apart into free fatty acids, which taste bad and are one of the major causes of rancidity. Mishandling of the raw material before factory processing also promotes enzyme action. The enzymes that release free fatty acids are called lipases, and this kind of rancidity is called lipolytic rancidity. Rancid dairy is especially gross because it has a high portion of the short chain fatty acids that are powerfully bad tasting and are, in fact, partially responsible for the disgtuisting flavor and smell of vomit.
- OXIDATION. Another cause of rancidity occurs due to ultraviolet light or heat or metals and other chemicals contaminating the fat or oil. These cause oxidation reactions, and they affect the PUFA fatty acids first. The off flavor results from the fact that oxidation reactions can release free fatty acids from the triglyceride, just as enzymes can, however the freed fatty acids are also damaged chemically and can be very toxic. Rancidity resulting from oxidative release of fatty acids is called oxidative rancidity. Fish is very high in PUFA fats and both oxidative and lipolytic rancidity play a role in generating the rotten smells of old fish.
Is eating rancid food unhealthy?
Yes, in general.
If it were just enzymatic rancidity at play releasing otherwise normal fatty acids, the answer would be no. In fact, animal like vultures that seek out already killed prey seem to enjoy the released free fatty acids we find disgusting. But these animals are generally eating carcasses that are only a few days old at most and generally the parts they eat at this stage contain mostly oxidation resistant saturated fatty acids.
We don’t eat rotting meat, unless we’re living with a native Greenlander and enjoying Kiviaq (made of auks fermented in a seal skin). So when we are hungry enough to consider downing something rancid its generally going to old nuts or seeds that are high in polyunsaturated fats. Because these PUFA fats oxidize easily, if you’re getting an off flavor from nuts or seeds its unhealthy and best avoided.
BEWARE OF “NEUTRAL” FLAVOR
Some of the most toxic fats have no flavor at all, and thus we can’t rely on taste to warn us that an oil contains toxic, oxidized fats. In fact, Canola, Soy and the other RBD oils are marketed to restaurants based on their lack of flavor, meaning the chef can use the same oil regardless of the spices and other flavor profiles.
Harvard Gets It Wrong
If you care about your health, ignore Harvard and Yale–at least for now. While many leading MDs are waking up to how wrong we were to insist that saturated fat was unhealthy, these two schools are digging their heels deep in the 1950s-era dogma. Their recommendations are to avoid saturated fat as much as possible and get roughly 25% of daily calories from polyunsaturated fat-rich foods like vegetable oils. The only evidence that supports this position is statistical (they do not offer a plausible physiologic mechanism), and their statistical work is seriously flawed by wrong assumptions and confounding variables.
In 2015, scientists at the NIH analyzed autopsy slides that were made as part of a study done in the 1970s. The study compared two diets, one rich in liquid vegetable oils (high polyunsaturated fats) and the other rich in hydrogenated vegetable oil (high in trans and saturated fats). Believe it or not, they found the folks on the hydrogenated vegetable oils had fewer heart attacks and strokes than the people on the liquid vegetable oils.
Walter Willet, the Dean of the School of Public Health, dismissed this finding as “a historical footnote.”
What Made Me Realize Harvard Gets it Wrong
I read a PhD dissertation that explained how polyunsaturated fatty acids (PUFAs) can ignite free radical reactions in our bodies. Free radical reactions are really bad. After reading more about oil processing and PUFA oxidation, I realized everything I’d learned about fats in medical school was wrong, and that it was necessary to reverse my earlier position on good fats and bad. As a practicing doctor, making this shift has not been easy, because it goes against what most of my colleagues still believe.
This section defines some common scientific terms for those interested in more of the chemistry.
BEST METHOD OF DETERMINING OXIDATIVE STABILITY: Activated Oxygen Method or AOM, I think it’s heating to 100C with peroxide, and the longer it takes to get to a certain point, the more it resists oxidation.
RANCIDITY=any off flavor. Two reasons: free fatty acids released from trigylceride, producing an off flavor. And Partial oxidation of the fatty acids.
FREE FATTY ACID FLAVOR (from vegetable oil manual I downloaded):
The liberated free fatty acids have a distinct flavor and odor which are more disagreeable when the fatty acid chain length is shorter than 14 carbons
OXIDIZED FATTY ACID FLAVOR (From veg oil manual)
Hydroperoxides themselves have no flavor or odor but break down rapidly to form aldehydes, many of which have a strong, disagreeable flavor and odor.
From naturalproductsinsider.com
“Hydrolytic rancidity, also called hydrolysis or enzymatic oxidation, occurs in the absence of air, but with moisture present. This normally is accomplished through enzymatic peroxidation, where enzymes found naturally in plant oils (i.e., lipoxygenase, cyclooxygenase) and animal fats (i.e., lipase) can catalyze reactions between water and oil.
Another degradation process is microbial rancidity, in which micro-organisms such as bacteria, molds and yeast use their enzymes to break down chemical structures in the oil, producing unwanted odors and flavors. Water needs to be present for microbial growth to occur.”
OXIDATION: Double bonds in the fatty acid reacting with oxygen.
OXIDIZED: Double bonds in the fatty acid that have reacted with oxygen to generate reaction products, usually with toxic e
IODINE VALUE: How many double bonds are present on average in the triglycerides in the oil. Does not distinguish between mono and poly. Higher value represents more double bonds.
PEROXIDE VALUE: Peroxide determination is the most widely accepted method for oil flavor quality determination. Peroxides are the major initial products of lipid oxidation and are measured by techniques based on their ability to liberate iodine from potassium iodide or to oxidize ferrous to ferric iron. Their content usually is expressed as milliequivalents of oxygen per kilogram of fat. Peroxide values of 0.5 meq/kg or less generally are necessary for a high flavor score. Because of the transitory nature or instability of peroxides, the level of peroxides may not serve as a true indicator of the actual state of oxidative rancidity of the fat or oil. During the course of oxidation, peroxide values reach a peak, then decline
FREQUENTLY ASKED QUESTIONS
I read seed oils were bad because of the omega-6, why didn’t you mention that?
Because that idea is a bit of a distortion of the truth. I wrote about that here.
What is artisanal oil and why is it the best?
Oil extracted using the highest quality mechanical (cold pressed) process that is tasty and wholesome enough to consume as is. Artesanal oils do not require any further steps and are sold unfiltered and unrefined. These are the most nutritious oils.
Can I eat seeds, nuts or fish that are high in PUFA?
Yes. Nature doesn’t make bad fats, factories do. For one thing, the seed oils are concentrated forms of PUFA, while the seeds, nuts and fish are not. Plus, when it comes to nuts and seeds, it’s the refining process that causes toxins, as described under How It’s Made: Edible Oils, above. When it comes to fish, consuming it raw, poached steamed or gently cooked will protect the PUFAs (so just don’t overcook it), and the fish tissue itself contains antioxidants so there will be far less PUFA breakdown.
I heard peanut oil was high in PUFA, why is it listed as good?
Peanut oil is higher in PUFA than olive oil, but still healthy when unrefined. This is because it has much less PUFA than the other seed oils, and the peanut seed protects its PUFA properly with plenty of antioxidants. These antioxidants would be removed during refining, but remain while unrefined.
Sesame oil is also high in PUFA, why do you list it as good?
Unlike the Hateful 8 seeds used to make industrially extracted and refined oils, sesame seeds are a traditional source of oil generally used for flavor. They are different for two more reasons as well. 1) Sesame seeds are relatively higher in fat (and antioxidants that protect PUFA) than most of the Hateful 8 seeds, which means the extraction process does not require harsh treatment. 2) The oil is not refined, and so the abundant antioxidants, minerals and vitamins are still present. I do recommend caution with heating, meaning don’t use for extended high heat cooking. Finally, I recommend combining it with peanut (see above) for stir fry.
Flax oil is high in PUFA, why is that good?
Flax is high in omega-3. It’s not used often as a culinary oil. Flax oil is sold as a supplement and is generally unrefined. Definitely don’t cook with flax because the omega-3 in flax is more unstable than the omega-6 in sesame.
Pumpkin oil, good or bad?
Pumpkin seed oil contains about 50% PUFA (mostly the omega-6 linoleic acid), 25% monounsaturated (oleic acid) and about 20% saturated. This high percentage of delicate PUFA means it must be unrefined to be edible and also that it’s not good to cook with.
What about high-oleic sunflower oil, is it better?
It is better, but not good, and the same applies to all high-oleic seed oils including soy, canola and safflower.
What about soy or sunflower lecithin?
This question came up so often I created a post, here: https://drcate.com/are-products-with-soy-lecithin-safe-to-eat-like-chocolate-ice-cream/
What are your thoughts on Thrive Algae Oil?
Not a big fan. They say “Highest level of monounsaturated fats and 75% less saturated fat than olive oil and avocado oil* the fact they think less saturated fat is a good thing indicates they don’t really understand the chemistry.
Since this question was posed, the company appears to no longer be making oil.
What about skin products made with seed oils?
While PUFAs applied to your skin won’t build up in your body fat, damage mitochondrial or promote systemic inflammation, they won’t do as much good for your skin as products made with stable oils that are higher in mono and saturated fat. As far as refined versus unrefined when it comes to skin, there’s much less benefit from unrefined. While unrefined indicates a better product (and probably a higher pricetag) your skin can’t absorb a significant amount of the nutrients that refining would remove, so you’re not missing much.
Palm oil is showing up in more foods. How can I tell if it’s refined?
If it’s unrefined the ingredients usually say so because it’s much more costly and they want you to know. The tougher questions is HOW was it refined? High-heat refining can produce carcinogenic compounds. Right now, the best method is to go by cost. If the product containing palm is less expensive than similar products containing soy, canola etc, it’s probably best to avoid it. I know that’s not a very good answer, sorry!
Is MCT oil acceptable? It is derived from coconut oil, but distilled with heat and pressure. Is it toxic?
It’s ok. Not toxic because the saturated fats are very stable to all the processing. Less nutritious than cream/butter, especially grass-fed.
How long does it take to get the excessive PUFA out of my body?
The half life is 18 months, so 4-5 years until you’re clear. That’s how long it takes to become ‘fully fat adapted.’ Fortunately you don’t need to wait years or even months before you start feeling results. You will start to experience improved energy the first day you swap out toxic fats for healthy ones in your breakfast.
Sources:
Interesterification leads to elevated glucose: https://dx.doi.org/10.1186%2F1743-7075-4-3
Refining Steps: http://canola.okstate.edu/canola-info/canolaoilmeal/oilprocessing.pdf
From Carnivore Aurelius: Is Vegetable Oil Bad For You
At DrCate.com
- Seed Oils: Questions and Answers For Your Health
This Post Has 325 Comments
Note: Please do not share personal information with a medical question in our comment section. Comments containing this content will be deleted due to HIPAA regulations.


















Is there a simple test (blood, etc) to determine your current inflammation level?
Not exactly. There are tests and panels that are called “inflammatory” tests but the body’s inflammatory responses are localized and complex, and no single test or even panel of tests can capture them. Although dozens of for profit lab testing companies will tell you otherwise.
Would it be possible to list actual commercial examples of healthy oil products available on Amazon?
Hi Dr Cate! Love your work 🙂
Mustard Oil – is it OK?
If unrefined, yes. If refined, no.
Hello Dr Cate, I am a few weeks pregnant with my fifth child since 2014 and sadly overweight. If I start getting rid of PFAS in my body, will it negatively affect the baby, ie will the baby absorb the PFAS as my body expels it, both in pregnancy and breastfeeding? Especially if I slightly lose weight naturally due to snacking less on junk food?
Which of your books would you recommend reading first for pregnancy advice?
Eating better will always help you and your baby. What I would NOT do is intentionally try to loose a lot of weight while pregnant or breastfeeding.
Of all my books Deep Nutrition gives you the most specific advice for motherhood and pregnancy diets (and fatherhood).
Dr. Cate, what’s your opinion on studies such as the one in JAMA Intern Med May 2025 comparing butter and plant based oils intake and mortality https://pubmed.ncbi.nlm.nih.gov/40048719/?
Is this a case where special interest is using bad science and torturing the data to say what it wants it to say, misleading the public into making bad choices for the sake of profit over health?
Pretty much. For details of how they torture the data, read my article about this study, here: https://drcate.com/seed-oil-study-used-possibly-fake-data/
Thanks for sharing a useful blog on oils which are very important for human life. It is a learning topic. I learned a lot on refined vegetable oil and many. Still it is confusing regarding use of appropriate oil for different types of patient. Which oil is good for heart patient? I like your blog and also like organic products.
Your website does not include Pomace Oil, used at Bolay Kitchen. I like Bolay because it is gluten free, but I am concerned about consuming Pomace Oil. Do you have an opinion about it? Thank you for your great book Dark Calories that improved my life immensely!
All oils are naturally gluten free. Pomace oil is the lowest quality olive oil fit for human consumption. It is typically refined using chemical solvents and then blended with virgin or extra virgin olive oil to make it “suitable for consumption.” It’s not good for you, but it’s not as toxic a the Hateful Eight oils.
Hi Dr Cate. After hours and hours of trying to research information on cooking oils (and other information on health, diet, insulin resistance, etc.), I am thrilled beyond words to have found the books and articles you have published/posted. My question is actually related to “how to cook,” and is an “offshoot” of the area of your expertise, but I’m hoping you can shed some light on this topic. I am concerned about the toxins in cookware, especially non-stick pans, so I am focusing on cast iron and the seasoning of cast iron. It seems that all cast iron now comes with a pre-seasoning done at the factory through the polymerization of an oil. In the case of the dutch oven that I am looking at, the pre-seasoning has been done with “natural organic flaxseed oil.” I know you have said not to cook with flax, but does the polymerization process block the release of PUFA if I cook in this pan with one of the all-purpose good oils. Thanking you in advance for any information you may be able to offer.
Seems like it would wash off eventually!
Some of the Simple Mills cookies have “Organic Palm Shortening” as an ingredient. Is that the same this as palm oil and is it okay?
Are seed oils ok in body products, like lotions? I have one lotion with certified organic sunflower oil and a body butter that has grapeseed oil.
Hi Dr. Cate, I was wondering if Organic Cold Pressed Sunflower Oil is okay in a omega 3-6-9 supplement? The product is Omega 3.6.9 Oil Blend by Omega Nutrition. It also contains cold pressed: organic flax seed oil, Organic pumpkin seed oil, Organic sunflower oil, Organic extra virgin olive oil.
Is organic high oleic sunflower oil ok?
https://lm.facebook.com/l.php?u=https%3A%2F%2Fsimplemills.zendesk.com%2Fhc%2Fen-us%2Farticles%2F360020520713-Why-sunflower-oil%3Ffbclid%3DIwZXh0bgNhZW0CMTEAAR2bps3BjgVrP__kf0Ctu4q24-WhUvX7g0-E939Rdz1Dmeuda5hs3Z4ejMQ_aem_oKfVMkbu7n7lrawP-xNlkA&h=AT1yTHjfJhDxJCopbddVfHiglhHbuXIch48wA8tDQFyKxe6ZxHvU9haT3RmanHRyAJFNO0cafX1B1V8_wf2pyoM5dXT9d2rXh6x1WdhJVx4Zo_8rOos6RrWZoVJyjt3Dp8eTHDO-kqlsPtKRc0eF
no
would love to see cultured ghee(prepared traditional way) also to the mix 😉
I’ve added ghee to to my materials recently, including those in Dark Calories, but will include here too in the next update. Thank you for the reminder!
Hi Dr. Cate! I really enjoyed watching your recent interview with Ben Greenfield. I own a gluten-free, battered, deep-fried chicken tender food truck in Tennessee, and have been, since I opened, under the impression that the canola oil I’ve been using has been cold pressed, and therefore is healthy. It is expeller pressed, in truth, and I feel the responsibility to make some changes. I am on a personal journey to recover my health and discover my full potential, and cannot in good conscience continue to serve food that is bad for my customers. Can you make any specific recommendations for oil that Is best for deep frying? Fried food will always be fried food, and presumably not the best for you, but I would like to make the product I serve the “healthiest“ version of fried food that I can. I would GREATLY appreciate your input on this. Thanks so much in advance!
-Dan O
I applaud you for your good conscience. That’s very rare. I normally can’t answer every individual’s questions (fortunately, most are already asked and answered elsewhere on my site), but I make an exception for people like you who feed so many others and who care about doing the right thing!!
Can you get tallow? That is what McDoanlds used to use until the AHA made them use hydrogenated vegetable shortening.
Thanks so much for taking the time. I CAN get tallow, though it’ll just be a matter of cost and figuring out what other details I need to consider (ie. grassfed, organic, etc.). I’ll start figuring this out ASAP. Thanks again!
Dan O
Also, before I jump the gun and make the switch, I’d just like to run this past you. The local non-GMO canola oil I’ve been using is described as “expeller-pressed”, but also as unrefined, unbleached and not deodorized. From what I’ve read on your site, these three seem to be the culprits. Just in case they’re offering a product you haven’t yet encountered, here’s a link to what I’ve been using: https://mpsfarms.com/store/Non-GMO-Canola-Oil-p158542131
Though the cost of tallow is significantly higher, as I mentioned, I’m committed to offering the “healthiest” fried food I can. This is what you do, so I will take your verdict as gospel, and run with it. ? Thanks again. I know your time is valuable, and I’m grateful for you spending it to help me.
Dan O
Also, before I jump the gun and make the switch, I’d just like to run this past you. The local non-GMO canola oil I’ve been using is described as “expeller-pressed”, but also as “unrefined, unbleached and not deodorized.” From what I’ve read on your site, these three seem to be the culprits. Just in case they’re offering a product you haven’t yet encountered, here’s a link to what I’ve been using: https://mpsfarms.com/store/Non-GMO-Canola-Oil-p158542131
Though the cost of tallow is significantly higher, as I mentioned, I’m committed to offering the “healthiest” fried food I can. This is your area, so I will take your verdict as gospel, and run with it. ? Thanks again. I know time is valuable, so I really appreciate you spending yours to help me.
Dan O
Grass fed tallow would likely be prohibitively expensive but any tallow would work as far as oxidation goes. I wonder if switching to solid may be difficult due to the different fryer technology?? That’s what I’ve heard from chefs, anyway.
If that is indeed quality canola it is definitely a step up from the RBD. But it’s omega-3 is problematic.
I’m looking and searching for the issue with the Omega-3 in canola on your site, and I’m coming up short. I’ll do the reading, if you can give me a little direction. Thanks!
Visit https://drcate.com/pufa-project/ and search for “Canola” on that page.
This is an article where I touch on the issues with omega-3 generally. https://drcate.com/should-i-take-fish-oil-supplements-benefits-and-harms/
And in this article in the Napa Register
https://napavalleyregister.com/lifestyles/food-and-cooking/the-canola-blob/article
I also go into the chemistry in my books.
Hello, I have been buying Bragg’s Unrefined Olive Oil for many years. Recently Spectrum Naturals is selling an Olive Oil labelled “Unrefined”. I was buying Braggs from Azure Standard and they are discontinuing Braggs. I got this explanation from them:
Thank you for reaching out with your question. The description provided on our website, describes the processing specifically. Although we do not use the word “unrefined,” the description details an unrefined process.
“Our oil is pressed from organic, Mediterranean olives. There’s no chemical processing, no hexane or heating involved. The oil is third-party tested for all the attributes of purity, so we’re absolutely sure there are no steroidal hydrocarbons, pesticides or additives. During the pressing the processes the oil at ambient / room temperature. The ideal temperature for crushing and pressing the olives is between 19?C (66.2 F) to 21?C (71.6F). If the environment is too cold, the mill crushing area is heated to stabilize the temperature.
We buy in large quantities and pour the olive oil in our new, convenient, Azure Market jugs in order to save you money. Nothing is added – the oil is not modified or adulterated in any way. Azure’s everyday virgin olive oil is every bit as flavorful, and actually more pure, than many of the big name brands.”
They claim that the Olive Oil they are selling is treated as Unrefined, but it is not labelled as such. Is this an oil I should buy?
Thank you, Stacie
Looks pretty good!
From the article:
“(Sesame is better if combined with peanut or other stabler fat, details below)”
Dr. Cate,
Have you ever heard of Mary Enig’s oil blend? You make it by using 1/3 extra-virgin olive oil, 1/3 sesame oil, and 1/3 coconut oil. It’s good for frying, and I also like to use it as a base for homemade mayo.
Sounds delicious!
I like it. ? It’s from the classic book “Nourishing Traditions.”
Thank you Dr. Cate, your analysis on the seeded oils is helpful!
I’ve been using Flora pumpkin oil, sesame oil, and flax oil;
They are listed as “organic, cold pressed, and unrefined.”
They are “pressed in a state of the art, low heat, light and oxygen free environment.“
I am using them strictly as supplements and NOT as cooking oils.
Your opinion on their benefits for a senior male?
THis is very informative blog, very nice info i loved it
Hello, Thanks for your website. And for coming onto the Dhru Podcast.
Two questions, three (or four) actually: what about mustard seed oil (cold pressed), hemp and peach stone oil?
And is it ok to add some flax oil or macadamia oil to overnight oats to make the lipophile micro nutrients more bioavailable?
If these oil have been refined, even if cold pressed, they’re not virgin and I would recommend you don’t eat them. My next article that I am releasing later this week will discuss this in a bit more detail. Check back soon!
I am wondering about LIGHT TASTING OLIVE OIL vs EVOO. The ingredient list on light olive oil says REFINED OLIVE OIL and VIRGIN OLIVE OIL.
I have been using the light oil in recipes that call for vegetable oil, but wonder about any harm processing might do??
It’s a blend of lower grade refined oil mixed with better oil. That reduces the cost and the nutrition. It would fall into my category I call “OK but not great” with refined palm, etx.
now i/m wondering if cold pressed and/or expeller pressed sunflower oil could be an ok option if is not made with heat, chemicals or bleach?
The only sunflower oil not made that way is unrefined, aka virgin oil. It’s good for salad dressings and quick cooking applications, similar to sesame oil.
Thank you for sharing all your wisdom!
I have spent a fair amount of time and energy on these subjects. I learn a whole lot of new things that benefit my body well, I unlearn some things, and sometimes I need to revise my conclusions. To summarize, I can say I have followed much of your nutritional advice, but not all, the last two/three years, and I am in the best shape of my life in all measurable ways.
Long story short: I probably eat too little omega-6 in my diet. My diet is almost fully meat-based, with an occasional inclusion of chicken and pork cuts. Can you advise me on which (preferably proteins) I should include in my diet to achieve the right amount or balance of omega-3/6?
Thank you!
Best regards
Einar
Hi Dr. Cate,
On a recent visit to the UK, I found that most brands of bread in the supermarkets listed “Palm Oil” as an ingredient (and in some cases Rapeseed or Sunflower oil as well). Your table shows Refined Palm as a bad fat, but what about Palm Oil where no degree of refinement is mentioned?
Palm oil refining is a bit of a quagmire because its in flux. It’s improved since 2017, so overall could be considered “okay but not great.” The default is refined unless specified “unrefined.”
Thanks, Dr. Cate. Good to know.
I have been taking Udo’s Oil as a supplement for years. I order it only in the colder months and freeze half and refrigerate the other half always. I use only butter and extra-virgin olive oil in cooking and baking, occasionally avocado oil. I eat very few processed foods (make my own crackers, bread, cook almost all our meals from scratch, eat out rarely, NO fast food ever.) Is Udo’s Oil an acceptable supplement? According to Flora and Udo, the oils are extracted under optimal conditions, low light, cool temperatures
Those are all the right terms! Most people don’t need to supplement omega 6 or 9, and the value of omega-3 supplements is minimal too
In the Don’t Eat / Bad Fats column, we see
“Most protein bars”
“Most cereals and granola”
What are some exceptions to these two?
Visit my Shopping List page, I found a few and listed them there.
Thats very helpful thanks. I think I understand.
Seed oils contain delicate compounds which degrade to something bad for us from heat and processing.
So for a vegetarian like me best to cook with olive oil and consume seeds in their natural state.
Great to hear you on Megyn Kelly ?
Hi Dr Cate,
I had been diagnosed with irritable bowel syndrome-IBS. I started to use good oil and fat, such as coconut oil and animal fat (pig fat) but I have diarrhea after using it. I don’t know this was related to the food I ate or the IBS I got already. If you have answer for this, please let me know more.
Thank you and really appreciate your effort and kindness !
Any abrupt and significant change to your diet will shift your gut flora, and that is why some people do better with baby steps. But there are multiple factors involved and if its a persistent issue you can feel free to schedule an appointment with me to guide you through the steps to sort it out. https://drcate.com/make-an-appointment-with-dr-cate-2/
Great article. Never knew expeller expressed was a 2 step process. Reading your book “Fat Burn Fix”. Great explanation on how these oils drive insulin resistance and diabetes. Trying to get my patients to make the connection between diet, chronic inflammation and why their back hurts most of the time. Question though on Olive oil and avocado oil. Have followed Paul Saladino MD and Tucker Goodrich and both seem relate that most of those oils have been diluted or adulterated by adding other hatefull 8 oils and are somewhat questionable. Why take the chance and not just stick with animal fats? Same with unrefined oils in nuts. We are already overloaded so why take anymore in? I suppose if we look at it as taking it in a stepdown process it makes sense. If we look at things on an evolutionary basis we only ate these seasonally. And shouldn’t we consider the oxalate issues? Thanks for the great work!
Hi Dr. Gregg. Great collection of questions. I’ll give you the simple answer to why not just eat only animal fats. I’ll give you two big reasons that shaped my thinking on the subject and there are many more: 1) Pleasure. Animal fats are awesome but using plant products wisely makes them taste better. We need variety of flavor or we get bored with our relatively restricted US food supply. 2) While plant oils can be adulterated, our pork and chicken and fish are also pretty messed up thanks to CAFOs and that impacts their nutritional value and their flavor.
On the oxalates, I’m not convinced they are the dietary villains some make them out to be. That’s a much longer conversation, though.
Hi Dr. Cate,
What are your thoughts on cold pressed, unrefined, organic black seed oil used in dressings not for heat.
If you like the taste use it, otherwise find a quality oil with a flavor you enjoy.
This is a great post! Thanks Dr Cate
I have recently read the Fatburn Fix and implemented changes to my diet. I have noticed positive effects already but also that I seem to sweat less during my workouts. Is this to be an expected side effect of these nutritional and behavioral changes to my diet?
Definitely could be for sure. Has to do with “cellular smog” discussed on page 105 of The FATBURN Fix. Excerpt “When your mitochondria are working efficiently, they power your cells with an abundance of energy while producing very little waste, which comes in the form of heat and free radicals. You can think of this as cellular smog. When your mitochondria are working less efficiently, as they are when your diet and body fat reflect the fatty acid profile of vegetable oils, they produce less energy and significantly more cellular smog. The less efficiently your mitochondria generate energy, the more heat and free radicals they will release.
The heat is one reason some people feel overly warm with minimal activity. And the smog overall is the source of a thousand health problems.”
What about castor oil? I’m hesitant with it but I read an article that talked about health of castor oil.
What is your stance on MCT oil and bulletproof coffee that is now trending?
HI Leslie,
MCT is empty calories but not toxic.
Is organic palm fruit oil the your same category as the “Hateful 8?” Is it bad for you?
It’s high in sat fat, and if not refined, it’s good. Taste? Not for me.
Hi Dr. Cate, do you know about Shark Liver Oil? Is this type safe? I have taken this for years and health benefits seem to be great. I’m having trouble finding information about PUFAs in it.
What about rapeseed oil? Is it good or bad?
What about rapeseed oil?
You’re Great Doctor Cate! ? Is ARGAN OIL good for the skin?
If helpful, the specific product I have in mind is Super Coffee.
Dr. Cate,
Is MCT oil on the list of good oils/fats? My understanding is that it is a combination of two good oils (coconut and palm kernel), but wanted to confirm since I am fastidiously adhering to your recommendations and have been seeing results after a few months. Thank you!
Betsy – Saw this article, came here to search mustard oil, and your post came up. Thought you might also find it interesting:
https://www.degruyter.com/d…
Bulletproof bars are recommended on the foods list, but looking at the ingredients, the bars include “
Safflower or Sunflower Oil.” Avoid these bars?
Hi Dr. Cate,
What are your thoughts on the UC Davis study testing the content of randomly selected avocado oil brands from local grocery stores? They found that most contained other kinds of oils, though labelled as only avocado oil, or were oxidized.
https://www.ucdavis.edu/foo…
It is already difficult to trust food labels as they are filled with half-truths and marketing ploys. Do you have any recommendations for identifying items that are accurately displaying their contents?
Thank you for the work you do to help us be healthy!
Frank, I suggest testing for obesogen levels by going to http://www.vibrant-wellness.com ask Dr. Cate to order it for you.
Unrefined peanut is OK (see the yellow column “OK but Not Great” in the image which hopefully is visible on top of this page)
Metformin is not a bad drug for T2DM, I prescribe it often when folks need something. If you keep going the way you’re describing here, I would expect you won’t need it for long!
Plain (presumably refined) peanut is fine, just not as vitamin and such rich as unrefined, you got it 100% right.
So happy to hear this, thank you for sharing!
Hey Cate,
I see Sesame and Peanut oil is put under good fats.. But Sesame and Peanut oil contain 2g and 1g of Linoleic acid per
a single teaspoon respectively.
Aren’t we supposed to have only 2g of PUFA per day?? So just anything more than 1 teaspoon will be too much. How is this on the healthy list?
Looking for a Vitamin D drop, sold in Canada, for my under 2 year old that does not contain vegetable or safflower oil – any recommendations?
Good Morning Dr. Cate. I’m a 67 year old male and have been following your program for the past 4 months and lost 29 lbs so far from my starting weight of 340. I had a comprehensive blood panel test done this week so I could compare my post Fat Burn Fix change data. to my prior diet data points. My Prior diet Glucose data points going back to 2014 were as follows. 11/27/2014 – 103, 8/5/2016 – 112, 9/12/2017 – 86, 7/16/2018 -79 , 9/18/2018 -83,11/13/2019 -93, 5/14/2020 -120, 9/22/2020 -104. (Note in 2017 and 2018 I was on a similar Keto diet and working out regularly 3 times a week lifting weights in a fitness club. Between those test dates and May 2020, I was very sedentary due to 3 different surgeries and then the Covid 19 quarantine isolation.) During the past 4 months my exercise has been limited to walking and riding my bike.
Thank you so much! My LDL is also increasing from 77 to 102.
Two questions; What should I be doing to move past this apparent weight loss plateau I’m at. I need to lose another 90 to 100 lbs.? What might be a reasonable expectation for how much longer it may take for me to get my fasting Glucose level under 100.
Thanks for your time.
Frank
Hi Dr. Cate, I’ve just started learning about the problem with seed oils from listening to Vinnie Tortorich’s podcast and want to thank you for being on his show and sharing this info with us! But I have a question that I don’t see answered here and haven’t heard anything about. I have used mustard oil for a long time but only occasionally because I like the flavor that it adds but now I’m realizing that it’s a seed oil so trying to figure out if I should buy it again… I found one on Amazon that I was going to buy that’s organic and coldpressed and the bottle says, “ unrefined, without any chemical processing.” What do you think of mustard oil?
Game Changes was a nice display of showmanship, I wouldn’t call it science. ? Olive pomace is not good, but not as bad as soy oil. Beyond burger shouldn’t use seed oils, and until they stop I have to recommend against. Pea protein as with whey protein and all protein powders is best avoided if at all possible. Sat fat and mono are healthy, PUFA are the ones that cause problems when consumed at current levels and when refined. I hope that makes sense. Would love a glowing Amazon review BTW if you believe the book deserves it, doing that really helps!!!!!
The paperback should have an index.
I have worked with folks with eating disorders, yes. In one consult I would be able to tell if my approach can help.
Anything in the good column when unrefined is in the “ok” column when refined. I hope that helps!
Hi, Dr. Cate,
Is there a bread or two out there that doesn’t contain harmful oils? I appreciate it!
Dr. Cate, I’m paying it forward!!! My doctor asked me at my appointment today how I got such a big improvement in my weight/bloodwork in less than three months. I told her all about you, the hateful eight, and all your recommendations that I am following. She was so impressed, she asked me for your name because she wants to read your books to pass on the information to her other patients. In ten weeks, I went from being prediabetic to now having my A1C in the normal range. I cut my triglycerides almost in half, lowered my cholesterol and blood sugar levels and lost weight. I will be forever indebted to you for bring me back to good health. Thank you, thank you, thank you!!!
https://uploads.disquscdn.c…
Hi Dr. Cate,
After seeing you on Bill Maher, then poring over your website, followed by reading the Fatburn Fix, and then reading and re-reading chapter 7 of Deep Nutrition, I literally threw away $200 worth of food from my fridge and pantry and have done what I think is a pretty thorough job of eliminating vegetable oils from my diet.
To facilitate this, I make a half gallon or so of mayonnaise each week that I use as a base for everything from egg salad to homemade bleu cheese dressing. I would appreciate your opinions or comments on the three types of oils that I have used due to cost, but mostly flavor profile.
In the image are the peanut from Walmart, sesame from Target, and avocado oil from Costco. The sesame is extra-virgin, but the peanut and avocado just say 100% oil, so I know they are refined. You say refined peanut/avocado are “okay, not great.” Is that strictly because they are less nutritious? Or are they actually harmful with other deleterious effects one should worry about where PUFAs are concerned?
To be VERY clear here…I am NOT cooking with these, just making mayo. I don’t really care so much about the nutrients, as the mayo is just a carrier for the rest of my diet which consists of mostly leafy greens, vegetables, and pasture-raised beef, chicken, and dairy.
1. By far the mayo with just peanut oil tastes best. Amazing actually.
2. Per your advice elsewhere, I also make it with a 5:1 peanut:sesame mix. It’s a little nutty for my taste, but still good enough.
3. The avocado (even refined) is of course a little grassy tasting, which I don’t really like. However…
4. A 50/50 blend of the avocado with the peanut/sesame mix isn’t too bad.
So, (remembering I don’t care about the nutrition so much) are any or all of these safe? What I really want you to tell me is that the plain peanut is fine, because I love it. But, if I you feel strongly about it being better to use one of the other combinations, I’m okay with that.
Thanks,
Loved your books,
Write more of them please
Are there any brands that you can suggest that you trust?
Cold-pressed is better. But!! More importantly, the product needs to be unrefined and taste flavorful. Chemicals used during refining destroy the delicate omega-3 and render those once beneficial compounds highly toxic.
In Denmark we have a tradition for using cold pressed rapeseed oil, which I guess is canola oil. We are being told, though, that it has the same benefits as cold pressed olive oil. Is the difference in whether it’s cold pressed or not? Or should I in general stay away from any rapessed oil?
Thank you so much!
I am pregnant and trying to make sure I get enough quality omega 3s, especially during this first trimester when many foods sound repulsive. Help me select a cod liver oil supplement? Some have a Maxsimil processing technique and I know you will understand the science the technique’s impact on the oil. https://fullcircleprenatal….
Hi Dr. Cate, the Almond oil I purchased for cooking is labeled ‘Expeller Pressed’, it does NOT say ‘unrefined’. I assume that it means that the Almond Oil is refined because they would otherwise put it on the label (?). Almond oil is listed above as GOOD FAT, but is it still good when expeller pressed but not refined or should the GOOD OIL specifically be labeled unrefined ? (Same Q for Avocado Oil). I hope I’m not repeating, found only comments and answers on expeller pressed in connection with bad oils. Thank you.
Thank you! I didn’t know you could get tested like this on your own. I wasn’t sure which option to go with but I just decided to go for the one with the advanced lipid panel, figuring that I might as well get more of a comprehensive or in depth result for that part.
However, I also added in the ‘CRPhs’ separately. I assumed that this might be the best other indicator of inflammation in the body. I didn’t include homocysteine because I wasn’t sure if that was as reliable or likely as CRPhs for indicating inflammation (but I could still add in homocysteine or something else to my order…..I’m not yet sure how long I will wait to actually go in and get this done).
Since I am going to be drastically reducing my intake of vegetable oils and perhaps changing other parts of my diet, I thought I should get tested for those things that it might be most interesting to compare at a future date, as it relates to metabolic health, inflammation, etc. If there is anything else you think might be very valuable of adding to the test in that regard, please let me know. Thanks. https://uploads.disquscdn.c…
Hello Dr. Cate – Saw you on Bill Maher, so glad I did. I’m somewhere between Phase I (accelerated) and Phase II; I felt significantly better just cutting out the 3 Cs and 3Ss and have lost approximately 7 pounds during regular Phase !.
Quick question – How do you recommend using MCT oil? I believe you mentioned using it like macadamia nuts, as a less nutritional energy boost between meals if needed?
Not so quick question – do you have any guidelines or resources for someone who has a history of an eating disorder, treated decades ago, but is having an exacerbation as a result of the pandemic/lockdown? She is medically stable, working with her endocrinologist and GP and me (I’m a psychologist). She had already cut out the bad oils, is doing some restriction during the day (but feels awful), having a protein bar and coffee with MCT oil throughout the day; and eating salad and chicken for dinner. and eating salad and chicken for dinner. (I suggested she consider coconut oil instead). She took a look at Fatburn Fix, but it was a little overwhelming so she is going to look at your website; I also suggested Primal Kitchen Bars and thinking of pointing her to Bullet Proof Coffee. She has lost weight, but again, it is stable. Her thyroid levels are off as is her cholesterol, as is the case for people with a hx of eating disorders. I really believe that the restorative part of Part 1 could be extraordinarily helpful for her, while working along with me and her doctors. Was also wondering if a consult with you might be helpful when she is a little farther along with her restriction? Traditional nutritionists have not been helpful to her in the past, not surprisingly.
One unrelated thing – if you made an index for Fatburn, I would buy it in a heartbeat!
Thank you for all you do –
Hi Dr. Cate: I found Superfood Creamer by Laird (Hamilton) Superfood. The ingredients are: Coconut Cream, Organic Coconut Sugar, Aquamin (calcium from marine algae), Organic Gum Acacia, Organic Mushroom Extracts (Chaga, Lion’s Mane, Cordyceps).
What do you think?
I have been trying to find a healthy non-dairy source of coffee creamer that is as satisfying as half and half. I am trying to consume more plant based products (for ethical reasons), and I really enjoy creamer in my daily coffee habit! I stopped using the ones that added soy lecithin.
Thank you so much!
Yes you can buy labs direct without a doctors order, one such company is DirectLabs. I put a few panels together to help folks with this type of question, check out the link here:
https://www.directlabs.com/…
Hi Dr. Cate, I’m new to your website/books, but have been rapidly absorbing all the information and making some dietary changes already. I’ve also been looking into what I can get screened for when getting a cholesterol test besides the standard good/bad/triglyceride/etc. The places I contacted so far didn’t seem to know anything about HOMA-IR. And as for the two components of that measurement, fasting glucose and fasting insulin, I’m finding that getting these done isn’t so simple either.
The places I talked with did say fasting blood sugar would likely be typically included, but as for my inquiry about fasting insulin, they either didn’t seem so familiar with that or said I’d need to talk to the doctor about why I’d want to get that done. It sounded like they were curious why I’d want to know about my insulin resistance if I don’t think I have diabetes and am not overweight, and I’m not sure if just saying that I’m concerned about my metabolic health or that I’ve had a low HDL in the past really will suffice.
Anyways, I’m sure I can figure out how to get tested for whatever I would like to at some point if I find the right place to get it done. But if I just got the cholesterol, and fasting insulin glucose and insulin, would that really tell me very much about my metabolic health and how I’ve been affected by my past vegetable oil heavy diet? Or is there some other measurement that I can get screened for (or a different type of test) that might be interesting to know as well? Thank you so much.
Hi Dr. Cate, I’m new to your website/books (saw you on Real Time), but have been rapidly absorbing all the information and making some dietary changes already (like no longer consuming large quantities of vegetable oil). I’m sure there’s some others like me who would want to know what might be the main things we’d want to get screened for when we have a cholesterol test besides the standard HDL/LDL/triglyceride/etc. numbers? The couple places I’ve looked into getting this done at didn’t seem to know anything about HOMA-IR when I called to ask about that. From my understanding, you can basically get the same result by learning your fasting glucose and fasting insulin levels.
And I’ve called a couple places so far, which both sounded like they would do the fasting glucose as a standard part of the test. However, testing for fasting insulin is something they either didn’t seem so familiar with, or said I’d need to talk to the doctor about getting done. (I think they were curious why I would want to know about insulin resistance if I don’t think I have diabetes, which I didn’t really have a great answer for since I’m not much of an expert yet. I’m not overweight, but from what I’ve read so far it sounds like there’s other measures of metabolic health and a lot more to it than just that. I’ve only had my cholesterol done once or twice in the past and the main concern I believe was a low HDL).
Anyways, I’m sure I can figure out how to get tested for whatever I would like to at some point if I find the right place to get it done. But if I just got the cholesterol, and fasting insulin glucose and insulin, would that really tell me very much about my metabolic health and how I’ve been affected by my past vegetable oil heavy diet? Or is there some other measurement that I can get screened for (or a different type of test) that might be interesting to know as well? Thank you so much.
Right. From my understanding and after re-reading…
PUFAs, regardless of being refined or not, are unstable fatty acids, meaning they are naturally prone to oxidation because of their polyunsaturated nature (affinity to bond with oxygen and break, releasing free radicals). Oxidation leads to mitochondrial inefficiencies or damage, which leads to metabolic dysfunction, which can lead to hypoglycemia… and so on, spiraling downwards.
Again from my understanding, oil refinement solvents like hexane, and other chemicals like bleachers and deodorizers just accelerate the process and guarantee that they will become oxidized. We do need a small amount of PUFAs as you’ve stated (something like 2-5% of fat calories I believe), but NOT 80% of fat cals that PUFAs currently make up on national average.
Incredible how pervasive vegetable oils are after you begin to pay attention…
Thanks Dr. Cate!
Please read the section that discusses expeller pressed above, “THE MYTH OF EXPELLER PRESSED” it should answer that question. And the answer to your second question on baking is ‘yes.’
Brown is better than white, and wild is better than brown. Wild rice has a lower glycemic index (50s versus 60s) and fewer carbs (35 versus 50), so you can have a little more of it before your blood sugar and insulin might bump.
Hi Dr. Cate,
Quick question about “Michele’s Granola”. I emailed them about their use of “expeller-pressed non-GMO canola oil”. The response I received was:
“…we believe the expeller pressed, non-GMO canola oil in our product is the best choice in regards to health as well as taste. It’s very important to us to use canola oil that is not extracted with the solvent hexane, which can destroy all the health benefits in this polyunsaturated oil.”
Does this mean their canola oil is completely unrefined, and if so, would that make it a “good” oil? Or, could it still partake in a bleaching and deodorizing process even without a hexane refinement stage?
In a previous comment, you said “skip canola for baking, sorry, because omega-3 degrade quickly and form toxic compounds when exposed to heat”. Does this still hold true for the unrefined canola Michele’s uses for baking granola?
Many thanks!
Brad
Hi Dr. Cate,
Quick question about “Michele’s Granola”. I emailed them about their use of “expeller-pressed non-GMO canola oil”. The response I received was:
“…we believe the expeller pressed, non-GMO canola oil in our product is the best choice in regards to health as well as taste. It’s very important to us to use canola oil that is not extracted with the solvent hexane, which can destroy all the health benefits in this polyunsaturated oil.”
Does this mean their canola oil is completely unrefined, and if so, would that make it a “good” oil? Or, could it still partake in a bleaching and deodorizing process even without a hexane refinement stage?
In a previous comment, you said “skip canola for baking, sorry, because omega-3 degrade quickly and form toxic compounds when exposed to heat”. Does this still hold true for the unrefined canola Michele’s uses for baking granola?
Many thanks!
Brad
I’m going to make you think this through. The missing piece of info is that cold pressed canola (or any oil) can still be refined. So knowing that, which would you think is better?
I’m going to make you think this through. The missing piece of info is that cold pressed canola (or any oil) can still be refined. So knowing that, which would you think is better?
They do have some value for those who can’t burn body fat effectively and are chronic snackers who need an energy boost.
They do have some value for those who can’t burn body fat effectively and are chronic snackers who need an energy boost.
Dear Dr. Cate. Which is better. Cold pressed canola or refined peanut?
Dear Dr. Cate. Which is better. Cold pressed canola or refined peanut?
What about mustard oil?
What about mustard oil?
Thank you so much Dr Shanahan! I value your advise and have been listening to your interviews. I cannot wait to read your new book:)
Thank you so much Dr Shanahan! I value your advise and have been listening to your interviews. I cannot wait to read your new book:)
An unrefined oil is going to be richer in antioxidants and better for everything, but manufacturers need to pretend that’s not the case so they can sell the lesser product. Do buy the best you can afford — as long as you enjoy the flavor!
An unrefined oil is going to be richer in antioxidants and better for everything, but manufacturers need to pretend that’s not the case so they can sell the lesser product. Do buy the best you can afford — as long as you enjoy the flavor!
If you must take the supplement and can’t find one made with coconut, the amount of seed oil is unlikely to be significant enough to warrant stopping it. But astaxanthin is itself a PUFA and would degrade into toxins in a matter of weeks, so I highly doubt the supplement contains as much as it promises.
If you must take the supplement and can’t find one made with coconut, the amount of seed oil is unlikely to be significant enough to warrant stopping it. But astaxanthin is itself a PUFA and would degrade into toxins in a matter of weeks, so I highly doubt the supplement contains as much as it promises.
they look good! I hope they taste good. Would be the same as artesanal grapeseed, yes, and limited exposure to heat would be best.
they look good! I hope they taste good. Would be the same as artesanal grapeseed, yes, and limited exposure to heat would be best.
The astaxanthin supplement that I take has safflower oil as one of the ingredients. Since this is a seed oil, is it best to avoid? Thank you and maximum blessings to you Dr Cate!
The astaxanthin supplement that I take has safflower oil as one of the ingredients. Since this is a seed oil, is it best to avoid? Thank you and maximum blessings to you Dr Cate!
Is roasted sunflower seed butter ok to consume.
Is roasted sunflower seed butter ok to consume.
See comment here: https://drcate.com/list-of-…
And if they add rice bran oil, yes avoid.
See comment here: https://drcate.com/list-of-…
And if they add rice bran oil, yes avoid.
Awesome! Thanks for the response.
Awesome! Thanks for the response.
Thank you for the reminder. I started Phase 1 from the book a week ago and got so excited I regurgitated a number of questions here – before reading the FAQ, reading more details in the FatburnFix, and checking for answers to questions already posted.
Fortunately the post was marked as SPAM – but it may reappear – if it does – I’ll try to remove it and make sure my questions have not already been answered.
Thank you for the reminder. I started Phase 1 from the book a week ago and got so excited I regurgitated a number of questions here – before reading the FAQ, reading more details in the FatburnFix, and checking for answers to questions already posted.
Fortunately the post was marked as SPAM – but it may reappear – if it does – I’ll try to remove it and make sure my questions have not already been answered.
Thanks for your input. All refined oils can contain compounds with carcinogens, usually at very low levels. Palm oil used to be refined at 400 before the expose on Nutella, now many are more gently refined and therefore contain very low levels of the carcinogenic compounds, if any. Unfortunately the label won’t tell you these key details.
Thanks for your input. All refined oils can contain compounds with carcinogens, usually at very low levels. Palm oil used to be refined at 400 before the expose on Nutella, now many are more gently refined and therefore contain very low levels of the carcinogenic compounds, if any. Unfortunately the label won’t tell you these key details.
There is no reason quality olive oil should lead to atherosclerosis. Whether HDL / LDL goes up or down depends on numerous variables and is not the point. The point is to avoid heart attacks and strokes.
There is no reason quality olive oil should lead to atherosclerosis. Whether HDL / LDL goes up or down depends on numerous variables and is not the point. The point is to avoid heart attacks and strokes.
PLEASE READ THE FAQ SECTION ABOVE BEFORE POSTING YOUR QUESTION!
PLEASE READ THE FAQ SECTION ABOVE BEFORE POSTING YOUR QUESTION!
Palm oil should be in the bad oil list. It doesn’t feel properly digestible and it’s being put into more and more foods. It also has carcinogenic properties.
Palm oil should be in the bad oil list. It doesn’t feel properly digestible and it’s being put into more and more foods. It also has carcinogenic properties.
Hi Dr. Cate. As you’ve mentioned lowering LDL may not be good. I’ve read that olive oil not only lowers LDL but it also lowers HDL. Is this true? And if so why is olive oil OK or even good?
Thank you for your great work!
Hi Dr. Cate. As you’ve mentioned lowering LDL may not be good. I’ve read that olive oil not only lowers LDL but it also lowers HDL. Is this true? And if so why is olive oil OK or even good?
Thank you for your great work!
I have a follow up – a broader question/issue.
I was a Vegan for many years – I’m not now but I’m still not much of a meat eater. No beef at alll, but some pork and chicken and fish. It seems that eating animal fat/protein is a major component of this program?? What foods can I substitute in place of meats? Are soy-based meat alternatives (morningstar as an example) – ok to use? Or can I just continue with Tofu and beans as I have (with some white meats thrown in?) I’m assuming beans of most kinds are fine?
Also I’ve always had high cholesterol, even when I was a Vegan – no matter what I did. My wife is very concerned that adding fats to my diet I’m risking my health. I’ve asked her to read the information available on this site and book – but that isn’t going to happen. I also have friends who are very ‘healthy’, no meats, very few carbs, mostly vegetarians… they think I’m crazy going this route. I’m excited and willing – but it feels like I’m swimming upstream with weights on. Giving up sweets and carbs is tough enough – but having my social circle telling me this is unhealthy – makes it that much more difficult. I’m all in – but looking for anyone here to offer suggestions on how to get the help and support I need.
I have a follow up – a broader question/issue.
I was a Vegan for many years – I’m not now but I’m still not much of a meat eater. No beef at alll, but some pork and chicken and fish. It seems that eating animal fat/protein is a major component of this program?? What foods can I substitute in place of meats? Are soy-based meat alternatives (morningstar as an example) – ok to use? Or can I just continue with Tofu and beans as I have (with some white meats thrown in?) I’m assuming beans of most kinds are fine?
Also I’ve always had high cholesterol, even when I was a Vegan – no matter what I did. My wife is very concerned that adding fats to my diet I’m risking my health. I’ve asked her to read the information available on this site and book – but that isn’t going to happen. I also have friends who are very ‘healthy’, no meats, very few carbs, mostly vegetarians… they think I’m crazy going this route. I’m excited and willing – but it feels like I’m swimming upstream with weights on. Giving up sweets and carbs is tough enough – but having my social circle telling me this is unhealthy – makes it that much more difficult. I’m all in – but looking for anyone here to offer suggestions on how to get the help and support I need.
Have spent a couple days on Phase I and I have some specific dietary questions:
– I have some questions below on certain food types – would it be easiest just to refer to what can be eaten on a Keto diet – or is that more restrictive, especially in Phase I? For example – I was going to ask about Fava/black/etc beans – but on the Keto diet they have too many carbs… same with vegetables such as Parsnips.
— I ask mainly because the book states its ok to have a slice of bread if it is from sprouted wheat but the Keto diet doesn’t care about that – only the total carb count
– Mariannes’ Avocado Oil – didn’t specify on the bottle – but on their website it says: “the oil is water extracted from the avocado fruit without the use of harmful chemicals and solvents, much the same way olive oil is made. We take it one step further and refine the oil” where it said it is refined – so no good? (Seems difficult to find unrefined oils in general – outside olive oil)
– Corn – popped (coconut oil) ok? Corn on the cob?
– RXBAR looks ok -however I do notice they are fairly high in carbs – which are strange since here are the ingredients: Peanuts, egg whites, chocolate, natural flavors?, sea salt, cocoa (maybe ‘natural flavors’ is sugar???
– I found a sprouted wheat bread – but it wasn’t frozen – Alvorado St. Bakery, Ingredients: sprouted organic whole wheat berries, water, gluten, honey, barley malt, millet, rolled oats, wheat starch, sea salt, yeast, sprouted organic corn, sunflower lecithin (bad?), sprouted lentils, sprouted pinto beans. (17g carb – 2g fiber-15g carbs net (2 of the carbs are sugar – so the honey?)
— comparing this to the Exekiel bread’s ingredients: Sprouted Wheat, Water, Sprouted Barley, Sprouted Millet, Balted Barley, Sprouted Lentils, Sprouted Soybeans, Sprouted Spelt, Yeast, Gluten, Salt (15g carb-3g fiber-12g carb net)
>> Not much difference (honey in the first), but carb-wise – they are the same 15g-3gfiber=12
>>> So this in general is one of my confusions – in reading the book – I can eat the sprouted bread, but on a hardcore Keto diet the 13g carb would be too much. Is there a general rule around the FatBurn Fix diet and Keto around this – or Phase 2 more on the strict keto (I haven’t gotten there yet).
– nut butter(s) pure ground nuts, nothing added, is that ok in general? on a celery stick for a snack?
– non dairy milk? (Soy, Oat, Almond) instead of milk? Any concerns (I don’t care for milk!)
Thanks – very much enjoyed the interview on Bill Maher, though I would have enjoyed it more if he had let you speak more!!
Have spent a couple days on Phase I and I have some specific dietary questions:
– I have some questions below on certain food types – would it be easiest just to refer to what can be eaten on a Keto diet – or is that more restrictive, especially in Phase I? For example – I was going to ask about Fava/black/etc beans – but on the Keto diet they have too many carbs… same with vegetables such as Parsnips.
— I ask mainly because the book states its ok to have a slice of bread if it is from sprouted wheat but the Keto diet doesn’t care about that – only the total carb count
– Mariannes’ Avocado Oil – didn’t specify on the bottle – but on their website it says: “the oil is water extracted from the avocado fruit without the use of harmful chemicals and solvents, much the same way olive oil is made. We take it one step further and refine the oil” where it said it is refined – so no good? (Seems difficult to find unrefined oils in general – outside olive oil)
– Corn – popped (coconut oil) ok? Corn on the cob?
– RXBAR looks ok -however I do notice they are fairly high in carbs – which are strange since here are the ingredients: Peanuts, egg whites, chocolate, natural flavors?, sea salt, cocoa (maybe ‘natural flavors’ is sugar???
– I found a sprouted wheat bread – but it wasn’t frozen – Alvorado St. Bakery, Ingredients: sprouted organic whole wheat berries, water, gluten, honey, barley malt, millet, rolled oats, wheat starch, sea salt, yeast, sprouted organic corn, sunflower lecithin (bad?), sprouted lentils, sprouted pinto beans. (17g carb – 2g fiber-15g carbs net (2 of the carbs are sugar – so the honey?)
— comparing this to the Exekiel bread’s ingredients: Sprouted Wheat, Water, Sprouted Barley, Sprouted Millet, Balted Barley, Sprouted Lentils, Sprouted Soybeans, Sprouted Spelt, Yeast, Gluten, Salt (15g carb-3g fiber-12g carb net)
>> Not much difference (honey in the first), but carb-wise – they are the same 15g-3gfiber=12
>>> So this in general is one of my confusions – in reading the book – I can eat the sprouted bread, but on a hardcore Keto diet the 13g carb would be too much. Is there a general rule around the FatBurn Fix diet and Keto around this – or Phase 2 more on the strict keto (I haven’t gotten there yet).
– nut butter(s) pure ground nuts, nothing added, is that ok in general? on a celery stick for a snack?
– non dairy milk? (Soy, Oat, Almond) instead of milk? Any concerns (I don’t care for milk!)
Thanks – very much enjoyed the interview on Bill Maher, though I would have enjoyed it more if he had let you speak more!!
Dr Cate, what about La Tourangelle’s other oils. I have been reading about their process and still wonder if the avacado oil is ok.
Dr Cate, what about La Tourangelle’s other oils. I have been reading about their process and still wonder if the avacado oil is ok.
It’s good!
It’s good!
It’s on the list under cookies, not crackers. But thanks to this type of feedback I added language to clarify that I’m not recommending the crackers due to the sunflower oil.
It’s on the list under cookies, not crackers. But thanks to this type of feedback I added language to clarify that I’m not recommending the crackers due to the sunflower oil.
The whole cholesterol thing is upside down. Low cholesterol may not be good. I write about the role of PUFA and sugar in heart attacks and this link includes some of the reliable resources on the topic: https://bit.ly/DrCateABCRef…
The whole cholesterol thing is upside down. Low cholesterol may not be good. I write about the role of PUFA and sugar in heart attacks and this link includes some of the reliable resources on the topic: https://bit.ly/DrCateABCRef…
Refined peanut is ‘ok’ but not great. Deep frying in tallow is a great option!
Refined peanut is ‘ok’ but not great. Deep frying in tallow is a great option!
Same as regular sesame oil: caution with heat. (So tasty)
Same as regular sesame oil: caution with heat. (So tasty)
Udos would likely be better than the fish oil. But avoiding the seed oils naturally increases your omega-3 for reasons that have to do with oxidation so depending on your diet you may not need to supplement.
Udos would likely be better than the fish oil. But avoiding the seed oils naturally increases your omega-3 for reasons that have to do with oxidation so depending on your diet you may not need to supplement.
Hi Dr Cate! Thank you, thank you, thank you! Your advice is literally saving my life! Is peanut oil that is unrefined AND expeller processed okay or not? It’s the Whole Foods 365 roasted peanut oil I’m asking about.
Hi Dr Cate! Thank you, thank you, thank you! Your advice is literally saving my life! Is peanut oil that is unrefined AND expeller processed okay or not? It’s the Whole Foods 365 roasted peanut oil I’m asking about.
Thanks! Trying to buy your latest book but “out of stock” everywhere! So popular after you appearance on RealTime! I’m not giving up! I really need a restart of my body machine!
Thanks! Trying to buy your latest book but “out of stock” everywhere! So popular after you appearance on RealTime! I’m not giving up! I really need a restart of my body machine!
Why are Simple Mills crackers on the food list you provide a link to when their 3rd is ingredient is organic sunflower oil?
Thanks
Why are Simple Mills crackers on the food list you provide a link to when their 3rd is ingredient is organic sunflower oil?
Thanks
Hello Dr. Cate,
Sorry for reposting. I understand you can’t respond to every question, but allow me to try again. I very much enjoyed your segment on Real Time with BM last week. It was an eye opener and I am trying to read as much as I can of your blog and publications. If someone is bold enough to go on record and risk their reputation to defend their conclusion I am all in!
I live in Italy, and while have easy access to the most wonderful (and inexpensive) cold pressed extra virgin olive oils I am having trouble finding unrefined peanut oil (or similar) for frying. So I am wondering just how bad is standard peanut oil found on the grocery shelves. If you had to use a processed oil for deep frying is refined peanut oil acceptable? Or is a non virgin olive oil preferable? Any other suggestion? Apologies if you have already addressed this in an earlier response.
Many thanks,
DG
Hello Dr. Cate,
Sorry for reposting. I understand you can’t respond to every question, but allow me to try again. I very much enjoyed your segment on Real Time with BM last week. It was an eye opener and I am trying to read as much as I can of your blog and publications. If someone is bold enough to go on record and risk their reputation to defend their conclusion I am all in!
I live in Italy, and while have easy access to the most wonderful (and inexpensive) cold pressed extra virgin olive oils I am having trouble finding unrefined peanut oil (or similar) for frying. So I am wondering just how bad is standard peanut oil found on the grocery shelves. If you had to use a processed oil for deep frying is refined peanut oil acceptable? Or is a non virgin olive oil preferable? Any other suggestion? Apologies if you have already addressed this in an earlier response.
Many thanks,
DG
I have shared this article with so many people! Thank you! What about toasted sesame oil?
I have shared this article with so many people! Thank you! What about toasted sesame oil?
The right-most column refers to foods that often contain seed oils. Some protein bars don’t but most do, so it helps to get in the habit of reading labels.
The right-most column refers to foods that often contain seed oils. Some protein bars don’t but most do, so it helps to get in the habit of reading labels.
Hi Darryl, great question on Game Changers. Their entire health argument is based on the idea that saturated fat is unhealthy because it raises blood cholesterol. What if the notion that cholesterol is unhealthy is wrong? I think it’s wrong and I describe an alternative etiology of atherosclerosis in Deep Nutrition. Here’s some other doctors who have questioned the standard teaching as well: https://bit.ly/DrCateABCRef….
Pomace oil is the cheapest, lowest quality you can feed to humans. More info https://www.oliveoiltimes.c…
Beyond burger is highly processed and as you point out made with Hateful 8. I never recommend.
I’m not a fan of any protein powder unless a person has pancreatic failure.
Saturated and mono unsaturated should compose the bulk of our dietary intake. We do need poly but it should come from whole food sources or high quality unrefined oils that are not used for extended high heat cooking.
Hi Darryl, great question on Game Changers. Their entire health argument is based on the idea that saturated fat is unhealthy because it raises blood cholesterol. What if the notion that cholesterol is unhealthy is wrong? I think it’s wrong and I describe an alternative etiology of atherosclerosis in Deep Nutrition. Here’s some other doctors who have questioned the standard teaching as well: https://bit.ly/DrCateABCRef….
Pomace oil is the cheapest, lowest quality you can feed to humans. More info https://www.oliveoiltimes.c…
Beyond burger is highly processed and as you point out made with Hateful 8. I never recommend.
I’m not a fan of any protein powder unless a person has pancreatic failure.
Saturated and mono unsaturated should compose the bulk of our dietary intake. We do need poly but it should come from whole food sources or high quality unrefined oils that are not used for extended high heat cooking.
Hi Dr. Cate. Great stuff last Friday night. I’ve reviewed your material and was wondering about supplements like Omega-3 fish oils and Udo’s 3-6-9 blend of oils to help promote healthy brain and eye function. My eye doctor actually suggested 1500 mg daily of a natural fish oil to promote healthy eyes. Yes, no?
Hi Dr. Cate. Great stuff last Friday night. I’ve reviewed your material and was wondering about supplements like Omega-3 fish oils and Udo’s 3-6-9 blend of oils to help promote healthy brain and eye function. My eye doctor actually suggested 1500 mg daily of a natural fish oil to promote healthy eyes. Yes, no?
Online. My fave is La Tourangelle
https://latourangelle.com/p…
Online. My fave is La Tourangelle
https://latourangelle.com/p…
Healthy nut heirarchy:
Sprouted >> Raw >> Oil Roasted (must be a good oil) & Dry Roasted
Healthy nut heirarchy:
Sprouted >> Raw >> Oil Roasted (must be a good oil) & Dry Roasted
I just read your info on supplements and was surprised to see magnesium oxide. My reading says magnesium glycinate and cytrate are more highly absorbable by the body. Also having a high vitamin D deficiency I found Puritan’s Pride 10,000IU pills 5x a week keep my vitamin D levels right in the middle.
I just read your info on supplements and was surprised to see magnesium oxide. My reading says magnesium glycinate and cytrate are more highly absorbable by the body. Also having a high vitamin D deficiency I found Puritan’s Pride 10,000IU pills 5x a week keep my vitamin D levels right in the middle.
Wow, we watched Dr. Cate on Bill Maher and what a great eye opening experience. After years of buying into the seed oils we had switched to Olive and Avocado oils to make our our dressings. Little did we know that we were not using the right kinds. We have now located cold pressed and unrefined versions of both.
One question, you list most protein bars. What is it in protein bars that determines a bad protein bar from a good or decent one? I have always read the ingredients and looked for any chemicals, fake sugars, etc.
Thank you so much for all the information you have posted. We need to get the word out on the myths of oils.
Wow, we watched Dr. Cate on Bill Maher and what a great eye opening experience. After years of buying into the seed oils we had switched to Olive and Avocado oils to make our our dressings. Little did we know that we were not using the right kinds. We have now located cold pressed and unrefined versions of both.
One question, you list most protein bars. What is it in protein bars that determines a bad protein bar from a good or decent one? I have always read the ingredients and looked for any chemicals, fake sugars, etc.
Thank you so much for all the information you have posted. We need to get the word out on the myths of oils.
Hi Dr Cate, would I be correct in assuming that refined olive oil aka “light olive oil” as it’s sometimes marketed, falls around the same category as refined avocado oil, “ok but not great”? Wondering because it’s usually my go-to oil for bread making and such, but want to be sure if I should stop using it and look for a healthier alternative. I’ve found conflicting documentation on it, particularly as it compares to extra virgin olive oil. Any insight would be greatly appreciated
Hi Dr Cate, would I be correct in assuming that refined olive oil aka “light olive oil” as it’s sometimes marketed, falls around the same category as refined avocado oil, “ok but not great”? Wondering because it’s usually my go-to oil for bread making and such, but want to be sure if I should stop using it and look for a healthier alternative. I’ve found conflicting documentation on it, particularly as it compares to extra virgin olive oil. Any insight would be greatly appreciated
Hi, Dr. Cate. Sorry about the repeat post, but wasn’t sure if you saw my last post since there was no response. I’m a Pediatrician, and very interested in health, especially in light of rising obesity rates in kids and in the general population. First, I am eating mostly vegan (still cheat sometimes) after watching Game Changers. Not sure what you think about the science behind that (assuming you’ve seen the movie), or if you feel plant-based eating is just a lot of hype? Second, I get healthy prepared meals delivered, and a lot of them have “olive pomace oil” listed in the ingredients. Is this type of oil OK, even though I believe it’s chemically refined? Third, I was wondering your thoughts on Beyond Burger and other plant-based meats, as they have a lot of the unhealthy oils. Also, any concern with pea protein powder (Naked Pea) for a morning protein shake? Finally, are you saying that, overall, saturated fats are healthier than mono and poly unsaturated fats, or does it really depend on the type of oil used? Sorry to bombard you with so many questions, but I thank you for taking the time to answer, and am looking forward to reading your books. By the way, you were great on Bill Maher! Stay well, and thanks again!! Regards, Darryl
Hi, Dr. Cate. Sorry about the repeat post, but wasn’t sure if you saw my last post since there was no response. I’m a Pediatrician, and very interested in health, especially in light of rising obesity rates in kids and in the general population. First, I am eating mostly vegan (still cheat sometimes) after watching Game Changers. Not sure what you think about the science behind that (assuming you’ve seen the movie), or if you feel plant-based eating is just a lot of hype? Second, I get healthy prepared meals delivered, and a lot of them have “olive pomace oil” listed in the ingredients. Is this type of oil OK, even though I believe it’s chemically refined? Third, I was wondering your thoughts on Beyond Burger and other plant-based meats, as they have a lot of the unhealthy oils. Also, any concern with pea protein powder (Naked Pea) for a morning protein shake? Finally, are you saying that, overall, saturated fats are healthier than mono and poly unsaturated fats, or does it really depend on the type of oil used? Sorry to bombard you with so many questions, but I thank you for taking the time to answer, and am looking forward to reading your books. By the way, you were great on Bill Maher! Stay well, and thanks again!! Regards, Darryl
Where can I purchase unrefined Almond Oil or Mac nut for cooking?
Where can I purchase unrefined Almond Oil or Mac nut for cooking?
I’m sure you must discuss this somewhere, but what is the story with nuts and seeds. I try to be very conscious of what I eat, but nuts and seeds are a big staple of my diet and I see that many of them are roasted in these bad oils. Removing them from my diet would create a hole in my diet that I wouldn’t know how to fill…any recommendations on how I can shop for nuts and seeds responsibly?
Go back to butter! I know it sounds like I’ve lost my mind. But know that sorting out the cholesterol and heart disease connection is what drove me down the seed oil rabbit hole. Chapter 7 of Deep Nutrition and 6 and & of The FATBURN Fix provide more info. For online resources and doctors with similar takes on the subject click here: https://bit.ly/DrCateABCRef…
I too wished for an index. Max 2 cups coffee/tea per day (more is ok, but makes you more likely to get withdrawal headaches) soy beans are fine, tofu is good too, its just no a super duper healthy food like some make it out to be.
Which fishes are considered “fatty fish”?
Thanks! I’m not sure why Dr. Cate hasn’t responded. Maybe she missed mine, and I should repost in a couple days? Or, maybe I somehow offended her. ?
I hate stevia. It tastes so awful, I do not get how people like it. It’s worse than saccharin, the aftertaste and flavor.
I did just get your book—an index would be helpful. To see what you say about caffeine, soy, and certain foods.
Years ago when coconut oil was the new big thing, I substituted it for butter spreads and cooking. My cholesterol shot WAY up. Since it was the one thing changed in my diet I concluded that it was why. I stopped using it, went back to olive oil, and my cholesterol went down. I am not sure why this oil is so beloved, besides taste.
Great question about reversing the damage. I’ve been scouring this page looking for the answer
Would love to read her response to your great questions!
Hi Dr. Cate,
Super interesting segment on Bill Maher! I recently was introduced to Udo’s Oil (http://udoschoice.com/produ… which is essentially a plant based omega supplement. I see that it contains rice bran oil. I do believe all the oils in this are unrefined, but does the addition of rice bran mean this should be avoided? Thanks so much,
Kate
Hello Dr. Cate,
I very much enjoyed your segment on Real Time last week. It was an eye opener and I am trying to read as much as I can of your blog and publications. If someone is bold enough to go on record and risk their reputation to defend their conclusion I am all in!
I live in Italy, and while have easy access to the most wonderful (and inexpensive) cold pressed extra virgin olive oils I am having trouble finding unrefined peanut oil (or similar) for frying. So I am wondering just how bad is standard peanut oil found on the grocery shelves. If you had to use a processed oil for deep frying would you recommend sticking with a variety from olives (e.g. pomace) or is refined peanut oil acceptable? Apologies if you have already addressed this in an earlier response.
Many thanks,
DG
Hi Dr. Cate, I have high cholesterol so I was using Earth Balance as a butter substitute. Looking at the label it contains a number of the fats that you are recommending people not consume. Are there some better butter substitutes that you would recommend?
I have been using Earth Balance as a butter substitute because I have high cholesterol. You are recommending that people not consume some of the the key ingredients. Is their another butter substitute that you would recommend?
That’s a difficult question. I’m not quite at the point where I’ll recommend only animals that were fed a species appropriate diet, although that would be ideal but we simply don’t raise animals right any more and don’t produce enough. The chicken fat will be lower in PUFA than the seed oils, and more importantly it will be more stable. If you can afford free range, where they ate fresh greens, bugs and a few grains, that’s your best bet.
Hi Dr. Cate – thank you for the incredibly helpful info. I’m wondering your thoughts on chicken thighs (let’s say, organic). Too much PUFAs?
That’s ok as a supplement. I wouldn’t cook with it, though.
Sorry where are you referring to? I can’t find on this page.
The whole idea that cholesterol is what clogs our arteries is about selling products. Not based in science. More info in Chapter 7 of Deep Nutrition and some good videos on the topic here: https://bit.ly/DrCateABCRef…
Palm fruit oil is ‘ok’ overall the fatty acids are very stable but if’s often refined, which strips nutrients.
It’s good as a nutritional supplement but I wouldn’t cook with it due to high PUFA content.
Thanks, I’ll look for the SK at Target!
Thanks for the new info! I noticed on your list that sesame seed oil is ok…what about hemp seed oil?
Just went grocery shopping, and have read your advice. I came across products using Palm Fruit Oil touting themselves as a healthy choice. Your thoughts?
Hi, Dr. Cate. I’m a Pediatrician, and very interested in health, especially in light of rising obesity rates in kids and in the general population. First, I am eating mostly vegan (still cheat sometimes) after watching Game Changers. Not sure what you think about the science behind that (assuming you’ve seen the movie), or if you feel plant-based eating is just a lot of hype? Second, I get healthy prepared meals delivered, and a lot of them have “olive pomace oil” listed in the ingredients. Is this type of oil OK, even though I believe it’s chemically refined? Third, I was wondering your thoughts on Beyond Burger and other plant-based meats, as they have a lot of the unhealthy oils. Also, any concern with pea protein powder (Naked Pea) for a morning protein shake? Finally, are you saying that, overall, saturated fats are healthier than mono and poly unsaturated fats? Sorry to bombard you with so many questions, but I thank you for taking the time to answer, and am looking forward to reading your books. By the way, you were great on Bill Maher! Stay well, and thanks again!! Regards, Darryl
I came to roughly the same conclusion a few years ago on my own after struggling for decades with health and weight. I decided to cut all food out of my diet that had added sugar, salt, flour or vegetable oil. I just needed to reset how I perceived food and this simple rule ended up being life changing. I lost 80lbs, was able to taste and enjoy simple foods again and generally got a lot healthier and stronger. But not before decades of miseasting took a toll.
Two questions, can the damage ever be undone and how many years before those inflammatory conditions reset?
How much “cheating” is ok? I still find myself having to eat things from time to time that aren’t my first choice, like at restaurants with friends or meals with family. I don’t want to be a jerk about it.
How would you classify the expeller pressed, unrefined seed oils produced by this company? Would they also be included in your hateful 8 or under the same category as artisanal produced grapeseed oil?
http://www.wholeheartedfoods.com
MCT won’t help you lose weight. It can help curb appetite, if that’s an issue. Vitamin D and supplement recommendations here: https://bit.ly/DrCateSupple…
Several brands make an avocado oil mayo now, including Chosen Foods, Primal Blueprint and Sir Kensington–my favorite. More brands & shopping guide info here: http://bit.ly/DrCatesShoppi…
See same question posted below, with my reply!
You list Stevia…have looked at Dr. Susan Swithers’ (Purdue Univ) research on it?
I’ve started to use this as well and wonder as well.
I daily take a few tablespoons of pumpkin seed oil. It is extra virgin, cold pressed. However, the label states that there are 8g of polyunsaturated fat per 15 ml. Also, 2.5g of saturated fat and 3g of monosaturated fat. Is this a healthy oil? thanks.
Algae oil?https://uploads.disquscdn.c…
Hi, Dr. Cate. I’m a Pediatrician in Chicago, and very interested in health, especially in light of rising obesity rates in kids and in the general population. First, I am eating mostly vegan (still cheat sometimes) after watching Game Changers. Not sure what you think about the science behind that (assuming you’ve seen the movie), or if you feel plant-based eating is just a lot of hype? Second, I get healthy prepared meals delivered, and a lot of them have “olive pomace oil” listed one the ingredients. Is this type of oil OK, even though I believe it’s chemically refined? Third, I was wondering your thoughts on Beyond Burger and other plant-based meats, as they have a lot of the unhealthy oils. Also, any concern with pea protein powder for a morning protein shake? Finally, are you saying that overall, saturated fats are healthier than mono and poly unsaturated fats? Sorry to bombard you with so many questions, but I thank you for taking the time to answer, and am looking forward to reading your books. By the way, you were great on Bill Maher! Stay well, and thanks again!! Regards, Darryl
If you had to pick one kind of mayonnaise (with or without egg), what would it be? Or, top three.
I am a middle-aged male who only recently began taking a spoonful of MCT oil every morning. Do you have an opinion on the effectiveness of it as a daily supplement? Also, several blood tests taken over the past year revealed widely different levels of Vitamin D; last year it was in the normal range, but the last one was very low. Possibly because I used to run outside everyday until a different work schedule sharply curtailed my jogging routine. Is a daily Vitamin D pill enough to make up for it?
What oil can you bake with?
Hi – saw you on Bill Maher’s show. Two ? s – 1) How do you rate Smart Balance – the blend with olive oil? And I’ve heard extra virgin is not so good for you, but honestly, it’s what I like to use and use a fair amount. 2) I suppose you think Hellman’s mayonaise is no good….? I don’t use much but hard to find Vegonaise…especially now in shopping online.
When it comes to sugar, unlike these Hateful 8, it’s not inherently toxic — the dose of sugar makes the poison. I recommend no more than 1 Tbsp total per day. If you need more than 1 tsp in your coffee, your taste buds are deadened to sweetness and slowly cutting back can wake them up again. If you go through that sugar detox, you’ll find you can enjoy the natural sweetness present in other foods much more. Try it!
While unrefined olive oil is the BEST, only light olive oil is something I’d avoid. The rest is good or at least ‘ok’. Avocado and coconut that have little to no flavor are refined, and even if refined they’re still “ok” not bad. It’s really the hateful 8 that we need to look out for and avoid. I hope that helps!
Thank you for this healthful information! After reading I immediately looked at the labels of my avocado, coconut and olive oils for the “unrefined” label….none. A brief search online (Amazon, VitaCost & Fred Meyer) proved to be more daunting than I anticipated! So now I’m on the hunt for acceptable healthy versions of these oils, preferably bottled in glass or metal that I can recycle. Are you aware of a good site and/or guide for keeping up with healthy oil manufacturers?
Is there anything acceptable that I can put in my morning coffee to make it sweet?
Is non-hydrogenated margarine, such as Becel, better than butter? I’m having a hard time with this. Thanks.
Thanks 🙂
Hello Dr Cate! I’ve seen an increase of healthy/organic/whole 30 products like salad dressings recently using “HIGH OLEIC SUNFLOWER OIL”. My basic 2 minute search on High Oleic tells me these are highly concentrated in monounsaturated fats rather than poly. A bit surprising, but I am curious if you have looked into this at all, and what your thoughts are on this. Thank you!
Oxidation is what we want to avoid, yes. Your total daily fat intake might be 100-150 grams, and if you consume fat sources other than olive oil, like dairy for example, you’re going to be doing pretty good.
Thanks Dr Cate. Being Greek, my experience with olive oil (10% PUFA like lard) is it will oxidize within 12-24 months in a green (light protected) glass jar sitting on dark shelf. Isn’t oxidation in the body what we want to avoid?
Also, if our body fat is 2% PUFA, how does our body deal with the remaining 8% PUFA in lard and olive oil?
Bacon fat and lard would have slightly different fatty acid profiles, but they are still reasonably low in PUFA and experience minimal to no industrial processing so the PUFA is reasonably well protected from oxidation.
I thought the whole point of algae oil was to get omega-3? I say that because you don’t want to cook with an oil that’s high in omega-3. They would need to publish their fatty acid profile for me to comment intelligently, though, and since it’s completely synthesized in a lab they should know that and share it.
A) more of an issue, but still an issue because it may select for undesirably skin bacteria
B) Not much absorption through skin for the longer chain fatty acids but fragments and short chains may get into bloodstream, more info here: https://www.nature.com/arti…
It means the oils were refined mechanically by centrifuge or pressure instead of chemically. Unrefined is always going to be better quality but usually very small portions of oil are produced this way by any given brand
if it’s truly glycerine however it may not be, according to this it’s allowed to contain fatty acids and when made from soy or corn etc it’s likely to be PUFAs: https://www.ams.usda.gov/si…
Hi Dr Cate,
1. What is your recommendation regarding skincare and hair products—moisturizers & leave-in conditioners—that contain things like Castor Oil/Vegetable oils?
A) Is it only an issue when you are in direct sunlight?
B) Are PUFAs absorbed through skin (even if not heated) still bad?
2. What is a general rule to know when PUFAs safe to consume or apply?
Hi Dr. Cate, What are your thoughts on using Algae Oil for cooking? I’ve attached a photo of the brand I buy. It’s advertised as having the highest amount of Monounsaturated fats. Is this just branded as healthy or is it actually safe to cook with at high heat? https://uploads.disquscdn.c…
Thank you for your awesome books. I am confused about lard. As “bacon from industrially produced pigs contains a lot of omega-6”, why is lard recommended at all? It has 5x the linoleic acid as in human bodies, causing build-up. Same for olive oil. It would seen only tallow is truly safe.
Pmega-6 is not the issue. I finally wrote an article about that here: https://drcate.com/omega-6-linoleic-acid-is-not-what-makes-seed-oils-bad/
I found a bottle of avocado oil where the label says “Expeller pressed, Naturally Refined”. So what does Naturally Refined mean? Is it the same as Refined, i.e. are Refined and Naturally Refined the same process? If not what is the difference?
I have the same question. Anyone has an answer please?
Should we be worried at all about vegetable glycerin?
It’s like sesame oil in that its fairly high PUFA so ideally would only be heated briefly and with spices or veggies that offer antioxidant protection, or with another fat that has more saturated and monounsaturated fatty acids.
Lectins are not present in the oil in any significant quantity. So that’s easy. But as to the PUFA profile yes its a borderline case and more of a judgement call so others may fall on the other side from me. I call ‘safe’ since unrefined peanut oil is so readily available (compared to say unrefined canola) and the flavor of unrefined peanut oil is so strong (and flavor equals nutrition), and since peanuts have so much vitamin 3 and other antioxidants.
It only took 9 months, but I believe I answered your question https://drcate.com/are-prod…
High oleic would definitely be superior to regular, however the oil would need to be unrefined and I’m not sure how you can make that determination unless the label specifies, therefore I doubt the manufacturers use unrefined oils if they don’t declare so on the label.
It’s not for cooking. Like flax it would be a nice source of omega-3 if unrefined and kept cold.
Hi omega – 3 oils should not be used for cooking, generally. YOu may be able add some to another oil, like peanut for example, because sometimes they stablize each other. For example, sesame oil is high pufa but added to peanut oil for stir frying the antioxidants in good quality sesame oil help protect the PUFA and the monounsaturated fatty acids keep the pufa protected during the short time on the heat.
Sure, ghee is a shade less healthy than butter because the minimal amount of proteins present are removed and the heating can theoretically break down PUFAs. Just be gentle.
It may have a little less vitamin E content, but it sure is yummy ! I use it for salads myself.
i use home made ghee for all kinds of cooking. Is that ok…
Where does hemp seed oil fall in this list?
Dr. Cate – Assuming soy lecithin is no good, being it’s only a dry (dried) product sourced from the original processed oil?
Dr. Cate I don’t understand the peanut oil and the peanuts. Peanut oil has 5000 times more omega6 and lots of linoleic acid. The peanuts themselves are loaded with harmful lectins.
Dr. Spiteller mentions linoleic acid frequently and James DiNicolantonio had the 2018 landmark publication where they have a box listing 29 ways the omega 6 oils are responsible for the heart epidemic, and mention linoleic acid frequently: “Omega-6 vegetable oils as a driver of coronary heart disease: the oxidized linoleic acid hypothesis” ( they referenced Dr. Spiteller 10 or 11 times in that).
If you think you are wrong on this one no big deal, to me.
As far as the lectins a doctor from Australia, Dr. Paul Mason, has now two youtube videos on the lectins, even though at first he scoffed at the idea. ( then he looked in the medical journals and is now applying this in addition to the low carb and keto regimen he recommends for many of his patients, with success )
I found one publication that checked 7 lectins for the ability to follow the vagus nerve into the brain, and 4 of them had this ability. These plants are often very determined to kill us!
Your book was wonderful and I reference it frequently and mention it in discussions frequently!
Hey Dr I want to know is mustard oil is bad for us or not.
I recently bought your book and didn’t find about mustard oil.
Seeds are healthier when raw or sprouted, but super yummy when cooked and although the nutrition is down a little they’re not going to be toxic unless you burn them. I have not heard of smoked cod liver oil as a process so thank you for bringing that to my attention. If its helping him, keep using it. For an whole food alternative you’d have to go with liver from another animal for the vitamins, and plenty of oily fish, like pickled herring, salmon and sardines.
Not going to be toxic due to sturdiness of the saturated fatty acids, not going to be good due to the refining that strips away nutrients. Its “ok”
HI Nicola, The rapeseed is the same as Canola, sorry to be the bearer of bad news. And they’re buying in to the whole saturated fat is bad idea, which is false and used as the excuse for selling cheap industrially refined oils. How many calories from fat are there?
Dear Dr. Shanahan, I love this oatmilk that I drink, but it has rapeseed oil in it 🙁 Is that the same as Canola? Also, is it definitely bad for me or does it depend on how processed/refined the rapeseed oil is?
They state “We think rapeseed oil is a good choice for our products since it contains both fatty acids and a high proportion of monounsaturated fat (compared to coconut oil, which is a saturated fat). We use only non-GMO rapeseed oil, processed as per other refined vegetable oils, but hexane is not involved in the processing of our oil. By the way, the rapeseed oil in our US Barista Edition is pressed and contains 0.0g/100g cholesterol and <0.1g/100g trans fat. Which means we can declare 0g on the package."
Thank you! Nicola
Dear Dr. Shanahan
Thank you very much for all the information you provide. I’ve just finished your book and really enjoyed it.
I do however have a few questions that so hope you can answer for me.
I make stone age bread/crakers (from seeds, water and salt only) and I make toasted pumpkin seeds for my family (sometimes toasted in duck-fat or lard). My kids really enjoy both as a replacement for chips etc. Is this OK? Or would the PUFAs in the seeds be oxidized?
Also – my husband has had lifelong problems with his sleep cycle and our Dr. tested him and found that his cortisol levels are out of wack. But since he started eating smoked cod liver (canned) twice a week the sleep problems have pretty much disappeared. We decided to supplement him w. cod liver after establishing that taking A, D and E-vitamins helped his sleep problems, but opting for a whole-food option instead of taking vitamin pills. But is the smoked cod-liver oxidized? And is it OK to eat the cod-liver oil from the can? The canning and smoking heats up the liver, but the oil does not smell bad at all (I sometimes use it on my salad).
Hope you can help. Thank you for everything you do!
Best Regards,
Dawn Hoff
Dear Dr. Shanahan,
I love all that you do and I am learning so much from all of your interviews. ( The REal Skinny on Fat) When you get a chance to answer as I know you are busy, what do you think of fractionated coconut oil?
Sincerely,
Rosanna
I’m a co-owner of a pasture-based family farm (Weathertop Farm in VA). I just listened to your podcasts (actually 2 back-to-back) on Primal Blueprint. I listened twice to make sure I heard right. I have long known about Omega 6 to 3 ratios and vitamins etc…, but if I heard right, when pasture is a significant percentage of the hogs diet, you said the ratio of saturated to unsaturated fat changes. I assumed most animals were like beef where diets didn’t change the molecular structure (double vs. single bonds), just the health of that type of fat. I’m very interested in knowing more of what goes into the process of changing the percentages of saturated to unsaturated fat in pork. I know you’re very busy, but I would greatly appreciate a little direction as to where to find research on this and some explanation of what the grass in the diet is doing in a non-ruminant animal. Many thanks, Cedric
HI Cedric! As is true with humans, the PUFA content of a pig’s diet is reflected in its body fat. There’s a decent amount of research in several species, look for “fatty acid profile essential dietary fats adipose” in google scholar. The exception to this trend seems to be cows, who’s complex gut flora may be why they’re more capable of preventing excessive PUFA concentration in their adipose. If I said pigs graze on grass I should have corrected myself, obviously that’s not the case.
HI Cedric,
The PUFA content in adipose reflects the PUFA content of an animals diet. Cows seem to be a bit of an exception. Lots of research can be found googline “fatty acid profile adipose dietary.” Sorry about the confusion around pigs eating grass, if I said that I misspoke–obviously they are not grazers. I may have been trying to say pasture raised, meaning access to outdoors.
Thanks so much for getting back to me. I think I’m getting closer to my source of confusion. I was not under the impression that you were claiming pigs were solely grazers. (Although with good rotation practices, I think one can get their grass intake to be as high as 25% of their diet…to see our hogs “grazing” see: https://www.facebook.com/WeathertopFarmLLC/videos/1053378281403141/ )
And please believe me, I am not trying to play gotcha or anything…but in Primal Blueprint Podcast #216, starting around 21:30 you start advising against bacon if sourced from CAFO’s. You go on to say 100% grain fed, confined pork is “…extremely high in PUFA’s, higher than…properly fed pigs…Fatty acid profile of beef doesn’t change that much when the cow is fed grain…but of pigs it does. It just goes to that high PUFA…and unhealthily high…”
I understood that to say grass intake could influence % of saturated fat to PUFA’s. I knew it changed the ratio of Omega-6 to O-3, but O-3 IS a PUFA. And simply changing the O-6 to O-3 ratio does not technically change the amount of total PUFA. Does that make sense? What am I missing? Can 20-25% intake of pasture change % of unsaturated fat to saturated fat?
Sorry I did not explain very well The answer to the question: Can 20-25% intake of pasture change % of unsaturated fat to saturated fat? Is yes–at least if we’re talking about replacing corn and soy feed with grass.
Great. Thanks for the clarification. Appreciate your time.
I recently heard about perilla oil and tiger nut oil. Do you recommend them?
Hi Dr. Cate,
Is it a bad idea to take a sunflower lecithin supplement for phosphotidylcholine? Is that just essentially taking sunflower oil (processed) ? Is there a better way to supplement PC? thanks!
Yes. And yes. If you can eat egg yolks you don’t need to supplement.
Hi, Dr Shanahan,
I have been eating a lot of plantain chips, either from whole foods or trader joe’s over the last year because they are AIP compliant.
WF Ingredient list: Plantains, Palm olein oil, and salt
TJ’s Ingredient List: Plantains, Palm olein
I realize the fatty acids in these chips, once fried, are likely of zero nutritional value, but I was always under the impression that palm oil or even palm olein oil (despite processing) was safe to eat because it was not an oxidizing oil. Is this an incorrect assumption, and should I stop eating these?
Regards,
Nick
I would very much like to know the answer to this as well! I have considered baking them from scratch but I can’t imagine they’d taste as good. I read somewhere that palm olein is toxic once oxidized, which I assume it is since the chips are fried?
They’re OK not good not bad.
AVAILABLE PETROLEUM PRODUCTS – D2, D6, JP54 & JETA1.
WE HAVE AVAILABLE PETROLEUM PRODUCTS FROM RELIABLE REFINERY IN RUSSIAN FEDERATION WITH BEST PRICE AND QUALITY.
BELOW PRODUCT ARE AVAILABLE WITH BEST OFFERS – FOB CI DIP AND PAY.
JP54: Quantity: 500,000-2,000,000 Barrels
D2: Quantity: 50,000-150,000 Metric Tons
D6 Virgin: Quantity: 400,000,000-800,000,000 Gallon
SERIOUS BUYERS PLEASE CONTACT US FOR MORE DETAILS WITH YOUR SPECIFIC REQUIREMENTS SO AS TO ENABLE US PROVIDE YOU WITH OUR WORKABLE PROCEDURE.
Sincerely.
Maksim Yaroslav (Mr.)
EMAIL: neftegazconsultant@yandex.ru
EMAIL: neftegazconsultant@mail.ru
Skype: neftegazconsultant
TEL: +7 9265036551
Dr. Cate,
Is this oil good or bad?
http://threefarmers.ca/camelina-oil/
I recently bought the 500 mL bottle with gold letters. Should I throw it out? Thank you for your important work!
Hi Dr. Cate,
Love your work! I saw that sunflower oil is listed as an oil to avoid. Should people also avoid sunflower butter and sunflower seeds?
Thanks!
Sunflower oil is a problem because of the refining, and of course sunflower seeds are not refined. The answer on sunflower butter is determined by if you make yourself at home and don’t bake anything–good–or if you’re buying a processed product–not good.
Sunflower oil is a problem because of the refining, and of course sunflower seeds are not refined. The answer on sunflower butter is determined by if you make yourself at home and don’t bake anything–good–or if you’re buying a processed product–not good.
Thank you Dr. Shanahan!
I currently buy the brand ‘Once Again’ organic unsweetened, unsalted sunflower seed butter because I don’t have time to make it myself. Is this a safe product to eat?? Thank you for your time.
There are large numbers of companies making all kinds of snack crackers these days. Everthing with GMO’s to kale and real sugar and and and. But virtually all of them still use oils from the BAD list.
Is there anything out there these days for chips and snacks from the supermarket we can buy that have good oils in them? Seems like there is a market out there for something that tastes great and is “better for you” because they have no “bad oils” they were processed with, or as ingredients.
I keep looking…. Thanks.
Health food stores often sell small local brands that are made with healthy oils, so that’s a great place to start looking. Best of luc!
Will patiently wait for an answer on the megatrans in Fish Oil question….
Thanks for your patience, see below!
Dear Dr Cate,
Still curious about Fish Oil and megatrans – in particular whether megatons are present in all omega 3 marine oils (krill oil, cod liver oil etc ). I note on pg.86 on Deep Nutrition you cite a study which showed that cod liver oil taken by pregnant mothers had protective and long lasting effects on the baby’s intelligence. So cod liver oil is not all bad. But does it, at the same time as offering these benefits, also put megatrans into our bodies? If so, what should we do?
Correct it’s not all bad. There is the potential for all of these marine oils and indeed all omega-3 supplements to have the degraded, Megatrans bad fats. But there is a lot of variability in the processing that can make a huge difference in the shelf life.
The higher quality supplemental oils generally use words like cold pressed and unrefined on the label.
It would be nicer if these companies could explain how they avoid oxidation during their processing.
This feels like a deeply significant piece of information. I’m just an ‘ordinary consumer’ – somewhat more informed after reading Deep Nutrition, but not an expert on fish oil processing methods, avoidance of oxidation etc. When buying fish oil, cod liver oil, krill oil etc it sounds like it is something of a lottery – there is no realistic way for me to know whether the oil I am buying contains Megatrans or not. (And it is is probably the case that most fish oils do contain it, but I will never know for certain which ones do or don’t.). So it would seem reasonable to conclude that our health would be better served by avoiding the stuff altogether?
Yes, we can be fooled into believing that because something’s in a capsule it’s all honkey dorey. You can sometimes tell by flavor but you would have to know what’s normal. If there’s an alternative you can use that would be better but you simply may not need it if you are not eating veg oils and your diet contains grass fed fats or a lot of PUFA rich nuts. Or oily cold water fish.
I just warned consumerlab.com about this. Hopefully they will do some private testing of fish oil supplement oxidation.
A quick question about Fish Oil .On page 168 of Deep Nutrition you mention that fish oil contains megatans and should be avoided. Does this apply to all omega 3 oils? For example are cod liver oil, fermented cod liver oil (e.g. by Green Pastures) and krill oil (as recommended by Dr Mercola) all equally suspect in this regard (that they contain megatrans)? Grateful for your advice as I am perplexed about this!
Kevin, YES it does apply. Sorry it took so long to get you the answer !!
fatty fish and printing out the list.
1) why do you say that it´s important to be cautious with heat when preparing fatty fish? I don´t eat raw fish…
2) is it ok to print out the above list of good fats and oils versus bad?
regards, b.i.g,
Please print yes and thank you for asking. Fish fats are also high in fragile PUFA fats, and heat destroys them. (thanks for your patience…)
I’d really like to know why Sunflowe and Safflower oils, specifically, are considered bad vegetable oils. Sunflowers themsleves are touted as having seversl health benefits. I am actually not quite sure what a Safflower is ?. Anyway, is it the refining process that makes these oils toxic? Is a small amount (as in the amount in some health foods sauces/dressings ok? Specifically I am vegetarian and recently fell in love with Beyond meats Beyond burgers. I see no major red flags except Sunflower oil, and refined coconut oil. But is this harmful if I were to eat these once or twice a week? Or is it just better to flag those ingredients like HF corn syrup and gluten and completely avoid. I do realize this is something processed, but the ingredients are not terrible and they are delish and a solid source of plant protein.
PUFAs are unstable and heating and processing damage them. Lots of detail in Chapter 7 and 8 of Deep Nutrition
Thank you for this info and research! I am wondering about nut butters, specifically my favorites: cashew and almond?
If you make them yourself, absolutely so. If its processed and you’re buying it, might be less good than yours depending on how its made, specifically how high its heated, but at least you won’t have the oil refining steps which are the worst, most chemically damaging.
Excellent – Thank you Dr. Cate!
I’m really enjoying Deep Nutrition audiobook Dr. Cate! I’m already recommending it to friends and family.
I do have two questions on specifics. My prepackaged, moist ready rice contains canola and/or sunflower oil. As far as I can tell, it amounts to 1-2g of fat. Is this amount (of oils) enough to worry about? I do enjoy the convenience of the packages.
On Endurance Planet podcast (if I heard correctly) you mentioned not eating sugars or carbs before a workout. For convenience, many of us take 1 gel 20 minutes before a race starts. Could you let me know if you think that is a bad idea in terms of performance? Thank you!
Hi Dr. Cate, I am reading Deep Nutrition and it has been life changing in terms of my perspective! Can you please comment on whether fish oil is safe to take? I have been taking the brand SFH (Stronger, Faster, Healthier) for years.
Also — can you please direct me to the best “restaurant survival guide” you have? Thank you!
Hi Dr. Cate-
I’m a Family Practice NP and I’m SO with you on the vegetable oils! I steer my patients and my family away from processed foods. Once in a while I will come across store- bought chips or crackers cooked in avocado or coconut oil (no PUFA on label, but doesn’t say refined or not).. are those OK for “occasional use”?
and what about heating up seeds at low heat? I make an “overnight” chia pudding in almond milk with berries which I stove-top heat in the AM for 5-7 min at very low heat. Is that fine? or risky? What about baking them (like cookies with flax or hemp seed?)
By the way, most of my patients are really on board with increasing their healthy fats- they can’t believe they can leave the skin on the chicken! So we are making progress!
Once in a while I will come across store- bought chips or crackers cooked in avocado or coconut oil (no PUFA on label, but doesn’t say refined or not).. are those OK for “occasional use”? Yes
On the “chia pudding in almond milk with berries which I stove-top heat in the AM for 5-7 min at very low heat. Is that fine?” Yes low, moist heat and being still in the seed protects the PUFA, same applies for baking since relatively moist.
Just read Tufts University Health & Nutrition Letter: Unsaturated Fat Best for Heart in their May 2017 isdue
New study published in The BMJ November 2016
Unbelievable instead of butter or lard choose corn canola soybeans instead of coconut milk Soy milk
How do you answer them Dr.Cate? How do we fight back?
They can’t admit they were wrong. It’s up to the next generation of dietitians and MDs (and anyone else who wants to join us) to form new organizations that get it right. The only way to fight it to not be snobby about ‘brand name’ institutions like Harvard Tufts and so on and recognize that we need to wash out the old guard and make way for the new. I’m teaching medical students and they’re eager to hear the truth so they can do right by their patients.
As a massage therapist I have spent years with my hands immersed in lotions containing canola oil. Is this as dangerous as ingesting? Or is it not as damaging when only contacted externally?
It would only affect your skin and even that not much, if at all, since the outer layers are just dead cells.
I use KTC refined coconut oil, because the coconut smell and taste swamps with unrefined (It’s also more affordable)
According to their FAQ, “it is a refined oil where any impurities in the raw material have been removed and it is not hydrogenated. The refining process for coconut oil is a physical process. The purpose of refining is to remove any impurities present in the raw material to produce 100% oil. The oil itself is not affected by the refining process.”
Elsewhere they describe their refining process:
————- quote ————–
Production
Coconut oil is physically stripped from the husk and extracted by pressing (not chemical extraction). The oil is then refined. During processing the oil is filtered number of times as well as at the end of process before loading or packing. End product has very low moisture content and high temperatures during deodorisation can guarantee that it is microbiologically safe.
Temperatures used in process of crush and refining are lower than the normal cooking temperatures – below 80-110C.
Where processes require to use higher temperature 180-220C vacuum is used to reduce temperature impact.
All process aids are food grade and fully removed from the oil.
For bleaching refineries use natural absorbent (bleaching earth or bleaching clay) and active carbon similar to what is used to purify water. Name “bleaching” came from the fact that the same time oil become lighter in colour because of pigments are also absorbed by clay.
——————— unquote ————-
Does this process conform to your expectations, or is it better at all?
That’s pretty standard refining process, yes. The reason refined coconut oil is ‘limited use’ and not ‘avoid’ has to do with the fact that the fatty acids are very stable and not much damaged during the refining. What’s lost is many antioxidants, vitamins and minerals.
Is refined, bleached, and deodorized coconut oil ok?
Limited use only. (I added it to the graphic, thank you for bringing it up!)
The mostly saturated fatty acids in coconut are not going to be damaged by the harsh RBD processing the way the PUFA fats are damaged in vegetable oil.
Yes it is ‘OK’ not good and not bad, but in the middle there. The reason ? Because the sat fat in coconut oil is ‘sturdy’ and won’t be damaged. The refining in this case primarily removes antioxidants and some minerals, but honestly coconut oil doesn’t have that much of either to remove. It’s almost the junk food of healthy fats.
How do you determine avocado oil from refined avocado oil? Same for peanut oil. What to look for on the label. I use avocado oil from Chosen Foods.
The bottles should say virgin or refined. If not, post a photo of the product with a link to it’s source from the company, if available, and I’ll look into it.
Says cold pressed & naturally refined?
OK then that’s refined and it’s in the limited use category. Meaning it’s not expected to contain toxic fatty acids to any significant degree, but its also stripped of significant levels of nutrients (minerals, antioxidants) so it won’t be particularly healthy.
I have been using this exact same avocado oil for cooking, is it bad to use it with high heat then? What if i find virgin avocado oil then it would be ok to cook it with? or which one would be the best to cook it with? Peanut oil? Thank you so much for your amazing book it changed my life! <3
Avo, peanut and olive can be used for high heat, you just have to stir and watch more carefully than you would with lower heat cooking.
Usually the unrefined will specify “unrefined” somewhere on the label.